Cellular dynamics of distinct skeletal cells and the development of osteosarcoma
- PMID: 37229448
- PMCID: PMC10203529
- DOI: 10.3389/fendo.2023.1181204
Cellular dynamics of distinct skeletal cells and the development of osteosarcoma
Abstract
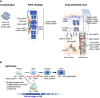
Bone contributes to the maintenance of vital biological activities. At the cellular level, multiple types of skeletal cells, including skeletal stem and progenitor cells (SSPCs), osteoblasts, chondrocytes, marrow stromal cells, and adipocytes, orchestrate skeletal events such as development, aging, regeneration, and tumorigenesis. Osteosarcoma (OS) is a primary malignant tumor and the main form of bone cancer. Although it has been proposed that the cellular origins of OS are in osteogenesis-related skeletal lineage cells with cancer suppressor gene mutations, its origins have not yet been fully elucidated because of a poor understanding of whole skeletal cell diversity and dynamics. Over the past decade, the advent and development of single-cell RNA sequencing analyses and mouse lineage-tracing approaches have revealed the diversity of skeletal stem and its lineage cells. Skeletal stem cells (SSCs) in the bone marrow endoskeletal region have now been found to efficiently generate OS and to be robust cells of origin under p53 deletion conditions. The identification of SSCs may lead to a more limited redefinition of bone marrow mesenchymal stem/stromal cells (BM-MSCs), and this population has been thought to contain cells from which OS originates. In this mini-review, we discuss the cellular diversity and dynamics of multiple skeletal cell types and the origin of OS in the native in vivo environment in mice. We also discuss future challenges in the study of skeletal cells and OS.
Keywords: bone marrow mesenchymal stem/stromal cells (BM-MSCs); cancer initiating cells; endosteal stem cells; lineage-tracing; osteosarcoma (OS); single-cell RNA sequencing (scRNA-seq); skeletal stem and progenitor cells (SSPCs); skeletal stem cells (SSCs).
Copyright © 2023 Otani, Ohnuma, Ito and Matsushita.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures
References
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Research Materials
Miscellaneous

