A randomized controlled trial to investigate the use of acute coronary syndrome therapy in patients hospitalized with COVID-19: the COVID-19 Acute Coronary Syndrome trial
- PMID: 37230416
- PMCID: PMC10204350
- DOI: 10.1016/j.jtha.2023.04.045
A randomized controlled trial to investigate the use of acute coronary syndrome therapy in patients hospitalized with COVID-19: the COVID-19 Acute Coronary Syndrome trial
Abstract
Background: Patients hospitalized with COVID-19 suffer thrombotic complications. Risk factors for poor outcomes are shared with coronary artery disease.
Objectives: To investigate the efficacy of an acute coronary syndrome regimen in patients hospitalized with COVID-19 and coronary disease risk factors.
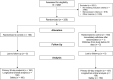
Methods: A randomized controlled, open-label trial across acute hospitals (United Kingdom and Brazil) added aspirin, clopidogrel, low-dose rivaroxaban, atorvastatin, and omeprazole to standard care for 28 days. Primary efficacy and safety outcomes were 30-day mortality and bleeding. The key secondary outcome was a daily clinical status (at home, in hospital, on intensive therapy unit admission, or death).
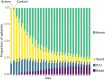
Results: Three hundred twenty patients from 9 centers were randomized. The trial terminated early due to low recruitment. At 30 days, there was no significant difference in mortality (intervention vs control, 11.5% vs 15%; unadjusted odds ratio [OR], 0.73; 95% CI, 0.38-1.41; p = .355). Significant bleeds were infrequent and were not significantly different between the arms (intervention vs control, 1.9% vs 1.9%; p > .999). Using a Bayesian Markov longitudinal ordinal model, it was 93% probable that intervention arm participants were more likely to transition to a better clinical state each day (OR, 1.46; 95% credible interval [CrI], 0.88-2.37; Pr [beta > 0], 93%; adjusted OR, 1.50; 95% CrI, 0.91-2.45; Pr [beta > 0], 95%) and median time to discharge to home was 2 days shorter (95% CrI, -4 to 0; 2% probability that it was worse).
Conclusion: Acute coronary syndrome treatment regimen was associated with a reduction in the length of hospital stay without an excess in major bleeding. A larger trial is needed to evaluate mortality.
Keywords: COVID-19 infection; anticoagulant agent; antiplatelet agent; ischemic heart disease; randomized controlled trial; thrombosis.
Copyright © 2023 The Authors. Published by Elsevier Inc. All rights reserved.
Conflict of interest statement
Declaration of competing interests P.K. receives research grants and consulting fees from Biosense-Webster, Abbott-Medical, Medtronic, and Boston Scientific. A patent for Ripple Mapping is licensed to Biosense-Webster, and royalties are paid to Imperial College. C.C. is the recipient of a British Heart Foundation Clinical Research Training Fellowship (number FS/20/14/34917). A.A. has had support to attend conferences from Bayer. A.N. received a research grant from the NIHR Academy. S.N. received payment or honoraria for speaker events/presentations from Bayer, Pfizer, and Philips and participates on an Advisory Board for Astra Zeneca. G.Y.H.L. is a consultant and speaker for BMS/Pfizer, Boehringer Ingelheim, and Daiichi Sankyo. No fees are received personally. N.M. is supported by a Chair Award, Program Grant, and Research Excellence Award (CH/F/21/90010, RG/20/10/34966, and RE/18/5/34216, respectively) from the British Heart Foundation. A.J.C. reports personal fees/consulting fees from Bayer, Pfizer, BMS, Daiichi Sankyo, and Boehringer Ingelheim; reports royalties and/or editorial fees from Oxford University Press, Wiley, and Springer Verlag; and participates on DSMBs/Advisory Boards at Biotronik, Johnson and Johnson, and Allergan. G.S.C. receives grants from the NIHR, participates on DSMBs, and is a nonexecutive director at the MHRA. W.G. reports fees for participation in Advisory Board from Amgen, Novartis, Pfizer, Principia Biopharma Inc—a Sanofi Company, Sanofi, SOBI, Grifols, UCB, Argenx, Cellphire, and Hutchmed; lecture honoraria from Amgen, Novartis, Pfizer, Bristol Myers Squibb, SOBI, Grifols, and Sanofi; and research grants from Bayer and BMS/Pfizer. M.T. reports Scientific Advisory Board personal fees from MorphogenIX and personal fees/honoraria from J&J/Actelion. R.A.-L. receives speaker’s honoraria from Philips Volcano, Medtronic, and Menarini. M.S.-S. has received honoraria from Pfizer (<£500). D.P.F., D.C., A.M., G.K., P.P., M.S., G.C., D.d.A.N., J.H., R.E., M.K., R.M., G.B., R.M.M., A.K., M.M., K.M., J.S., S.J.C., M.J.L., N.P., R.P., A.S.M.B., M.D., I.M., N.P., V.C. have no competing interests to disclose.
Figures
References
-
- Zhou F., Yu T., Du R., Fan G., Liu Y., Liu Z., Xiang J., Wang Y., Song B., Gu X., Guan L., Wei Y., Li H., Wu X., Xu J., Tu S., Zhang Y., Chen H., Cao B. Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: a retrospective cohort study. Lancet. 2020;395:1054–1062. - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials