Nanostructured TiO2 Arrays for Energy Storage
- PMID: 37241492
- PMCID: PMC10223775
- DOI: 10.3390/ma16103864
Nanostructured TiO2 Arrays for Energy Storage
Abstract
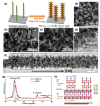
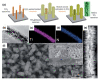
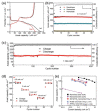
Because of their extensive specific surface area, excellent charge transfer rate, superior chemical stability, low cost, and Earth abundance, nanostructured titanium dioxide (TiO2) arrays have been thoroughly explored during the past few decades. The synthesis methods for TiO2 nanoarrays, which mainly include hydrothermal/solvothermal processes, vapor-based approaches, templated growth, and top-down fabrication techniques, are summarized, and the mechanisms are also discussed. In order to improve their electrochemical performance, several attempts have been conducted to produce TiO2 nanoarrays with morphologies and sizes that show tremendous promise for energy storage. This paper provides an overview of current developments in the research of TiO2 nanostructured arrays. Initially, the morphological engineering of TiO2 materials is discussed, with an emphasis on the various synthetic techniques and associated chemical and physical characteristics. We then give a brief overview of the most recent uses of TiO2 nanoarrays in the manufacture of batteries and supercapacitors. This paper also highlights the emerging tendencies and difficulties of TiO2 nanoarrays in different applications.
Keywords: TiO2; batteries; energy storage; nanoarrays; supercapacitors.
Conflict of interest statement
The authors declare no conflict of interest.
Figures












Similar articles
-
Ion-Exchange Loading Promoted Stability of Platinum Catalysts Supported on Layered Protonated Titanate-Derived Titania Nanoarrays.ACS Appl Mater Interfaces. 2019 Jun 19;11(24):21515-21525. doi: 10.1021/acsami.9b04378. Epub 2019 Jun 7. ACS Appl Mater Interfaces. 2019. PMID: 31132239
-
Recent progress in design, synthesis, and applications of one-dimensional TiO2 nanostructured surface heterostructures: a review.Chem Soc Rev. 2014;43(20):6920-37. doi: 10.1039/c4cs00180j. Chem Soc Rev. 2014. PMID: 25014328
-
Multi-functional silver nanoprism-titanium dioxide hybrid nanoarrays for trace-level SERS sensing and photocatalytic removal of hazardous organic pollutants.Spectrochim Acta A Mol Biomol Spectrosc. 2023 Sep 5;297:122701. doi: 10.1016/j.saa.2023.122701. Epub 2023 Apr 3. Spectrochim Acta A Mol Biomol Spectrosc. 2023. PMID: 37054569
-
Oxygen Vacancy Engineering in Titanium Dioxide for Sodium Storage.Chem Asian J. 2021 Jan 4;16(1):3-19. doi: 10.1002/asia.202001172. Epub 2020 Nov 23. Chem Asian J. 2021. PMID: 33150730 Review.
-
Synthesis and Electrochemical Energy Storage Applications of Micro/Nanostructured Spherical Materials.Nanomaterials (Basel). 2019 Aug 27;9(9):1207. doi: 10.3390/nano9091207. Nanomaterials (Basel). 2019. PMID: 31461975 Free PMC article. Review.
Cited by
-
Green biosynthesis of titanium dioxide nanoparticles incorporated gellan gum hydrogel for biomedical application as wound dressing.Front Chem. 2025 Apr 17;13:1560213. doi: 10.3389/fchem.2025.1560213. eCollection 2025. Front Chem. 2025. PMID: 40313399 Free PMC article.
-
Improved UV Photoresponse Performance of ZnO Nanowire Array Photodetector via Effective Pt Nanoparticle Coupling.Nanomaterials (Basel). 2024 Sep 4;14(17):1442. doi: 10.3390/nano14171442. Nanomaterials (Basel). 2024. PMID: 39269104 Free PMC article.
-
Core-Double-Shell TiO2@Fe3O4@C Microspheres with Enhanced Cycling Performance as Anode Materials for Lithium-Ion Batteries.Materials (Basel). 2024 May 24;17(11):2543. doi: 10.3390/ma17112543. Materials (Basel). 2024. PMID: 38893808 Free PMC article.
-
Comparative Analysis of Anodized TiO2 Nanotubes and Hydrothermally Synthesized TiO2 Nanotubes: Morphological, Structural, and Photoelectrochemical Properties.Materials (Basel). 2024 Oct 24;17(21):5182. doi: 10.3390/ma17215182. Materials (Basel). 2024. PMID: 39517457 Free PMC article.
-
Sintering Aid Strategy for Promoting Oxygen Reduction Reaction on High-Performance Double-Layer LaNi0.6Fe0.4O3-δ Composite Electrode for Devices Based on Solid-State Membranes.Membranes (Basel). 2023 Jun 15;13(6):603. doi: 10.3390/membranes13060603. Membranes (Basel). 2023. PMID: 37367807 Free PMC article.
References
-
- Banerjee S., Dionysiou D.D., Pillai S.C. Self-Cleaning Applications of TiO2 by Photo-Induced Hydrophilicity and Photocatalysis. Appl. Catal. B Environ. 2015;176–177:396–428. doi: 10.1016/j.apcatb.2015.03.058. - DOI
-
- Henderson M.A. A Surface Science Perspective on TiO2 Photocatalysis. Surf. Sci. Rep. 2011;66:185–297. doi: 10.1016/j.surfrep.2011.01.001. - DOI
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

