Safety, Tolerability, and Pharmacokinetics of β-Cryptoxanthin Supplementation in Healthy Women: A Double-Blind, Randomized, Placebo-Controlled Clinical Trial
- PMID: 37242207
- PMCID: PMC10222860
- DOI: 10.3390/nu15102325
Safety, Tolerability, and Pharmacokinetics of β-Cryptoxanthin Supplementation in Healthy Women: A Double-Blind, Randomized, Placebo-Controlled Clinical Trial
Abstract
Background: β-cryptoxanthin is a dietary carotenoid for which there have been few studies on the safety and pharmacokinetics following daily oral supplementation.
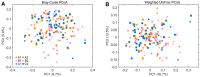
Methods: 90 healthy Asian women between 21 and 35 years were randomized into three groups: 3 and 6 mg/day oral β-cryptoxanthin, and placebo. At 2, 4, and 8 weeks of supplementation, plasma carotenoid levels were measured. The effects of β-cryptoxanthin on blood retinoid-dependent gene expression, mood, physical activity and sleep, metabolic parameters, and fecal microbial composition were investigated.
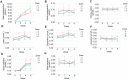
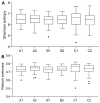
Results: β-cryptoxanthin supplementation for 8 weeks (3 and 6 mg/day) was found to be safe and well tolerated. Plasma β-cryptoxanthin concentration was significantly higher in the 6 mg/day group (9.0 ± 4.1 µmol/L) compared to 3 mg/day group (6.0 ± 2.6 µmol/L) (p < 0.03), and placebo (0.4 ± 0.1 µmol/L) (p < 0.001) after 8 weeks. Plasma all-trans retinol, α-cryptoxanthin, α-carotene, β-carotene, lycopene, lutein, and zeaxanthin levels were not significantly changed. No effects were found on blood retinol-dependent gene expression, mood, physical activity and sleep, metabolic parameters, and fecal microbial composition.
Conclusions: Oral β-cryptoxanthin supplementation over 8 weeks lead to high plasma concentrations of β-cryptoxanthin, with no impact on other carotenoids, and was well tolerated in healthy women.
Keywords: Asian women; carotenoids; fecal microbiome; gene expression; mood; physical activity.
Conflict of interest statement
The authors declare no conflict of interest.
Figures


References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

