Nonselective and A2a-Selective Inhibition of Adenosine Receptors Modulates Renal Perfusion and Excretion Depending on the Duration of Streptozotocin-Induced Diabetes in Rats
- PMID: 37242515
- PMCID: PMC10222446
- DOI: 10.3390/ph16050732
Nonselective and A2a-Selective Inhibition of Adenosine Receptors Modulates Renal Perfusion and Excretion Depending on the Duration of Streptozotocin-Induced Diabetes in Rats
Abstract
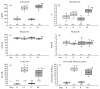
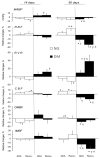
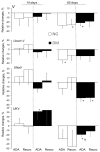
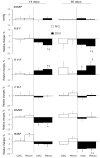
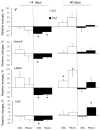
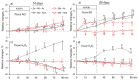
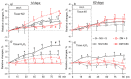
Long-lasting hyperglycaemia may alter the role of adenosine-dependent receptors (P1R) in the control of kidney function. We investigated how P1R activity affects renal circulation and excretion in diabetic (DM) and normoglycaemic (NG) rats; the receptors' interactions with bioavailable NO and H2O2 were also explored. The effects of adenosine deaminase (ADA, nonselective P1R inhibitor) and P1A2a-R-selective antagonist (CSC) were examined in anaesthetised rats, both after short-lasting (2-weeks, DM-14) and established (8-weeks, DM-60) streptozotocin-induced hyperglycaemia, and in normoglycaemic age-matched animals (NG-14, NG-60, respectively). The arterial blood pressure, perfusion of the whole kidney and its regions (cortex, outer-, and inner medulla), and renal excretion were determined, along with the in situ renal tissue NO and H2O2 signals (selective electrodes). The ADA treatment helped to assess the P1R-dependent difference in intrarenal baseline vascular tone (vasodilation in DM and vasoconstriction in NG rats), with the difference being more pronounced between DM-60 and NG-60 animals. The CSC treatment showed that in DM-60 rats, A2aR-dependent vasodilator tone was modified differently in individual kidney zones. Renal excretion studies after the ADA and CSC treatments showed that the balance of the opposing effects of A2aRs and other P1Rs on tubular transport, seen in the initial phase, was lost in established hyperglycaemia. Regardless of the duration of the diabetes, we observed a tonic effect of A2aR activity on NO bioavailability. Dissimilarly, the involvement of P1R in tissue production of H2O2, observed in normoglycaemia, decreased. Our functional study provides new information on the changing interaction of adenosine in the kidney, as well as its receptors and NO and H2O2, in the course of streptozotocin diabetes.
Keywords: adenosine receptors; hydrogen peroxide; hyperglycaemia; nitric oxide; renal blood flow; renal excretion; streptozotocin.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
References
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

