Design, Synthesis, and Evaluation of Niclosamide Analogs as Therapeutic Agents for Enzalutamide-Resistant Prostate Cancer
- PMID: 37242518
- PMCID: PMC10222209
- DOI: 10.3390/ph16050735
Design, Synthesis, and Evaluation of Niclosamide Analogs as Therapeutic Agents for Enzalutamide-Resistant Prostate Cancer
Abstract
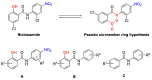
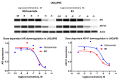
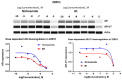
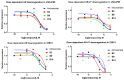
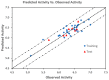
Niclosamide effectively downregulates androgen receptor variants (AR-Vs) for treating enzalutamide and abiraterone-resistant prostate cancer. However, the poor pharmaceutical properties of niclosamide due to its solubility and metabolic instability have limited its clinical utility as a systemic treatment for cancer. A novel series of niclosamide analogs was prepared to systematically explore the structure-activity relationship and identify active AR-Vs inhibitors with improved pharmaceutical properties based on the backbone chemical structure of niclosamide. Compounds were characterized using 1H NMR, 13C NMR, MS, and elemental analysis. The synthesized compounds were evaluated for antiproliferative activity and downregulation of AR and AR-V7 in two enzalutamide-resistant cell lines, LNCaP95 and 22RV1. Several of the niclosamide analogs exhibited equivalent or improved anti-proliferation effects in LNCaP95 and 22RV1 cell lines (B9, IC50 LNCaP95 and 22RV1 = 0.130 and 0.0997 μM, respectively), potent AR-V7 down-regulating activity, and improved metabolic stability. In addition, both a traditional structure-activity relationship (SAR) and 3D-QSAR analysis were performed to guide further structural optimization. The presence of two -CF3 groups of the most active B9 in the sterically favorable field and the presence of the -CN group of the least active B7 in the sterically unfavorable field seem to make B9 more potent than B7 in the antiproliferative activity.
Keywords: androgen receptor variant 7 (AR-V7) down-regulators; enzalutamide-resistant prostate cancer; niclosamide analogs; structure–activity relationship (SAR).
Conflict of interest statement
The authors declare no conflict of interest.
Figures



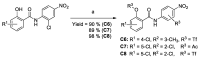


Similar articles
-
Niclosamide and Bicalutamide Combination Treatment Overcomes Enzalutamide- and Bicalutamide-Resistant Prostate Cancer.Mol Cancer Ther. 2017 Aug;16(8):1521-1530. doi: 10.1158/1535-7163.MCT-16-0912. Epub 2017 May 12. Mol Cancer Ther. 2017. PMID: 28500234 Free PMC article.
-
Isolation and characterization of castration-resistant prostate cancer LNCaP95 clones.Hum Cell. 2021 Jan;34(1):211-218. doi: 10.1007/s13577-020-00435-6. Epub 2020 Sep 20. Hum Cell. 2021. PMID: 32954481 Free PMC article.
-
Niclosamide suppresses cell migration and invasion in enzalutamide resistant prostate cancer cells via Stat3-AR axis inhibition.Prostate. 2015 Sep;75(13):1341-53. doi: 10.1002/pros.23015. Epub 2015 May 13. Prostate. 2015. PMID: 25970160 Free PMC article.
-
Current status of androgen receptor-splice variant 7 inhibitor niclosamide in castrate-resistant prostate-cancer.Invest New Drugs. 2018 Dec;36(6):1133-1137. doi: 10.1007/s10637-018-0653-2. Epub 2018 Aug 7. Invest New Drugs. 2018. PMID: 30083960 Review.
-
Prognostic Value of Androgen Receptor Splice Variant 7 in the Treatment of Castration-resistant Prostate Cancer with Next generation Androgen Receptor Signal Inhibition: A Systematic Review and Meta-analysis.Eur Urol Focus. 2018 Jul;4(4):529-539. doi: 10.1016/j.euf.2017.01.004. Epub 2017 Jan 23. Eur Urol Focus. 2018. PMID: 28753843
Cited by
-
Therapeutic Approaches to Targeting Androgen Receptor Splice Variants.Cells. 2024 Jan 4;13(1):104. doi: 10.3390/cells13010104. Cells. 2024. PMID: 38201308 Free PMC article. Review.
-
Successive paired electrochemical late-stage modification of niclosamide a common anthelmintic drug. A green protocol for the synthesis of new drug-like molecules.RSC Adv. 2025 May 29;15(22):17803-17810. doi: 10.1039/d5ra02025e. eCollection 2025 May 21. RSC Adv. 2025. PMID: 40443689 Free PMC article.
-
Valine-Niclosamide for Treatment of Androgen Receptor Splice Variant-Positive Hepatocellular Carcinoma.Cancers (Basel). 2025 Jul 31;17(15):2535. doi: 10.3390/cancers17152535. Cancers (Basel). 2025. PMID: 40805231 Free PMC article.
-
N-terminal domain of androgen receptor is a major therapeutic barrier and potential pharmacological target for treating castration resistant prostate cancer: a comprehensive review.Front Pharmacol. 2024 Sep 18;15:1451957. doi: 10.3389/fphar.2024.1451957. eCollection 2024. Front Pharmacol. 2024. PMID: 39359255 Free PMC article. Review.
References
-
- Hoang D.T., Iczkowski K.A., Kilari D., See W., Nevalainen M.T. Androgen receptor-dependent and -independent mechanisms driving prostate cancer progression: Opportunities for therapeutic targeting from multiple angles. Oncotarget. 2017;8:3724–3745. doi: 10.18632/oncotarget.12554. - DOI - PMC - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

