RadioLigand Therapy with [177Lu]Lu-PSMA-617 for Salivary Gland Cancers: Literature Review and First Compassionate Use in France
- PMID: 37242537
- PMCID: PMC10222097
- DOI: 10.3390/ph16050754
RadioLigand Therapy with [177Lu]Lu-PSMA-617 for Salivary Gland Cancers: Literature Review and First Compassionate Use in France
Abstract
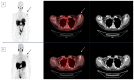
Salivary gland cancers are rare tumors comprising a large group of heterogeneous tumors with variable prognosis. Their therapeutic management at a metastatic stage is challenging due to the lack of therapeutic lines and the toxicity of treatments. [177Lu]Lu-PSMA-617 (prostate-specific membrane antigen) is a vectored radioligand therapy (RLT) initially developed to treat castration-resistant metastatic prostate cancer with encouraging results in terms of efficacy and toxicity. Many malignant cells could be treated with [177Lu]Lu-PSMA-617 as long as they express PSMA as a consequence of androgenic pathway activation. RLT may be used when anti-androgen hormonal treatment has failed, particularly in prostate cancer. [177Lu]Lu-PSMA-617 has been proposed in certain salivary gland cancers, though the expression of PSMA is demonstrated by a significant uptake using [68Ga]Ga-PSMA-11 PET scan. This theranostic approach could be a new therapeutic option, warranting prospective investigation in a larger cohort. We review the literature on this subject and offer a clinical illustration of compassionate use in France as a perspective for administering [177Lu]Lu-PSMA-617 in salivary gland cancer.
Keywords: [177Lu]Lu-PSMA-617; salivary gland cancer; vectored radioligand therapy.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
References
-
- Van Boxtel W., Uijen M.J.M., Verhaegh G.W., Willems S.M., Jonker M.A., PALGA Group. Schalken J.A., van Engen-van Grunsven I.C.H., van Herpen C.M.L. Prognostic value of PSMA, c-MET and E-cadherin in salivary duct carcinoma. Oral Oncol. 2020;110:105018. doi: 10.1016/j.oraloncology.2020.105018. - DOI - PubMed
-
- Aegerter D.P., Cosmidis D.A. Participants Bureau REFCOR. [(accessed on 19 September 2022)]. Available online: http://refcor.org/files/116/recommandations/refcor_glandes_salivaires.pdf.
-
- Airoldi M., Pedani F., Succo G., Gabriele A.M., Ragona R., Marchionatti S., Bumma C. Phase II randomized trial comparing vinorelbine versus vinorelbine plus cisplatin in patients with recurrent salivary gland malignancies. Cancer. 2001;91:541–547. doi: 10.1002/1097-0142(20010201)91:3<541::AID-CNCR1032>3.0.CO;2-Y. - DOI - PubMed
Publication types
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous