Effect of the Composition of Leuzea and Cranberry Meal Extracts on Metabolic Processes in Norm and Pathology
- PMID: 37242551
- PMCID: PMC10221024
- DOI: 10.3390/ph16050768
Effect of the Composition of Leuzea and Cranberry Meal Extracts on Metabolic Processes in Norm and Pathology
Abstract
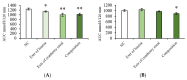
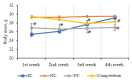
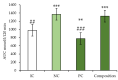
This study was conducted to evaluate the effects of long-term administration of a new herbal composition of leuzea and cranberry meal extracts at a dose of 70:500 mg/kg in healthy and pathological mice. After 4 weeks of daily composition administration to healthy CD-1 mice and C57BL/6 mice with diet-induced metabolic syndrome, oral glucose tolerance test (OGTT), serum biochemical examination and histology of internal organs were performed. Additionally, histological examination of white and brown adipose tissue was performed to evaluate the ability of the composition to prevent abdominal obesity in C57BL/6Ay (agouti yellow) mice. The results showed that the composition increased tissue sensitivity to glucose in healthy CD-1 mice; at the same time, it did not worsen the course of pathological processes in pathological mice. In both cases, the application of the developed composition was safe and contributed to the restoration of metabolic parameters.
Keywords: adipocytes; cranberry; ecdystene; extracts; leuzea; metabolic syndrome; mice; ursolic acid.
Conflict of interest statement
The authors declare no conflict of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript; or in the decision to publish the results.
Figures



References
Grants and funding
LinkOut - more resources
Full Text Sources

