Glycovaccine Design: Optimization of Model and Antitubercular Carrier Glycosylation via Disuccinimidyl Homobifunctional Linker
- PMID: 37242563
- PMCID: PMC10223089
- DOI: 10.3390/pharmaceutics15051321
Glycovaccine Design: Optimization of Model and Antitubercular Carrier Glycosylation via Disuccinimidyl Homobifunctional Linker
Abstract
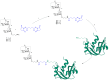
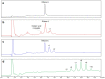
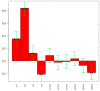
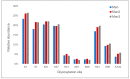
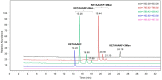
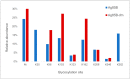
Conjugation via disuccinimidyl homobifunctional linkers is reported in the literature as a convenient approach for the synthesis of glycoconjugate vaccines. However, the high tendency for hydrolysis of disuccinimidyl linkers hampers their extensive purification, which unavoidably results in side-reactions and non-pure glycoconjugates. In this paper, conjugation of 3-aminopropyl saccharides via disuccinimidyl glutarate (DSG) was exploited for the synthesis of glycoconjugates. A model protein, ribonuclease A (RNase A), was first considered to set up the conjugation strategy with mono- to tri- mannose saccharides. Through a detailed characterization of synthetized glycoconjugates, purification protocols and conjugation conditions have been revised and optimized with a dual aim: ensure high sugar-loading and avoid the presence of side reaction products. An alternative purification approach based on hydrophilic interaction liquid chromatography (HILIC) allowed the formation of glutaric acid conjugates to be avoided, and a design of experiment (DoE) approach led to optimal glycan loading. Once its suitability was proven, the developed conjugation strategy was applied to the chemical glycosylation of two recombinant antigens, native Ag85B and its variant Ag85B-dm, that are candidate carriers for the development of a novel antitubercular vaccine. Pure glycoconjugates (≥99.5%) were obtained. Altogether, the results suggest that, with an adequate protocol, conjugation via disuccinimidyl linkers can be a valuable approach to produce high sugar-loaded and well-defined glycovaccines.
Keywords: Ag85B antigen; antitubercular vaccines; chemical glycosylation; disuccinimidyl linkers; glycoconjugate characterization; glycoconjugate vaccines; glycosylation optimization.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

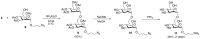


References
-
- Smith B.R., Guo Z. Oligosaccharide Antigen Conjugation to Carrier Proteins to Formulate Glycoconjugate Vaccines. Methods Mol. Biol. 2021;2183:305–312. - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources

