Modulation of Transcription Profile Induced by Antiproliferative Thiosemicarbazone Metal Complexes in U937 Cancer Cells
- PMID: 37242567
- PMCID: PMC10220753
- DOI: 10.3390/pharmaceutics15051325
Modulation of Transcription Profile Induced by Antiproliferative Thiosemicarbazone Metal Complexes in U937 Cancer Cells
Abstract
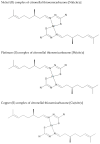
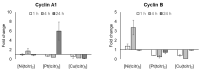
Since the discovery of cisplatin, the search for metal-based compounds with therapeutic potential has been a challenge for the scientific community. In this landscape, thiosemicarbazones and their metal derivatives represent a good starting point for the development of anticancer agents with high selectivity and low toxicity. Here, we focused on the action mechanism of three metal thiosemicarbazones [Ni(tcitr)2], [Pt(tcitr)2], and [Cu(tcitr)2], derived from citronellal. The complexes were already synthesized, characterized, and screened for their antiproliferative activity against different cancer cells and for genotoxic/mutagenic potential. In this work, we deepened the understanding of their molecular action mechanism using an in vitro model of a leukemia cell line (U937) and an approach of transcriptional expression profile analysis. U937 cells showed a significant sensitivity to the tested molecules. To better understand DNA damage induced by our complexes, the modulation of a panel of genes involved in the DNA damage response pathway was evaluated. We analyzed whether our compounds affected cell cycle progression to determine a possible correlation between proliferation inhibition and cell cycle arrest. Our results demonstrate that metal complexes target different cellular processes and could be promising candidates in the design of antiproliferative thiosemicarbazones, although their overall molecular mechanism is still to be understood.
Keywords: DNA damage response pathway; expression profile study; molecular action mechanism; ribonucleotide reductase; thiosemicarbazone metal complexes.
Conflict of interest statement
The authors declare no conflict of interest.
Figures



References
-
- Hannon M.J. Metal-based anticancer drugs: From a past anchored in platinum chemistry to a post-genomic future of diverse chemistry and biology. Pure Appl. Chem. 2007;79:2243–2261. doi: 10.1351/pac200779122243. - DOI
LinkOut - more resources
Full Text Sources

