Multi-channel recordings reveal age-related differences in the sleep of juvenile and adult zebra finches
- PMID: 37244927
- PMCID: PMC10224955
- DOI: 10.1038/s41598-023-35160-1
Multi-channel recordings reveal age-related differences in the sleep of juvenile and adult zebra finches
Abstract
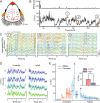
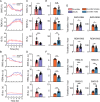
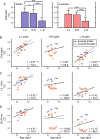
Despite their phylogenetic differences and distinct pallial structures, mammals and birds show similar electroencephalography (EEG) traces during sleep, consisting of distinct rapid eye movement (REM) sleep and slow wave sleep (SWS) stages. Studies in human and a limited number of other mammalian species show that this organization of sleep into interleaving stages undergoes radical changes during lifetime. Do these age-dependent variations in sleep patterns also occur in the avian brain? Does vocal learning have an effect on sleep patterns in birds? To answer these questions, we recorded multi-channel sleep EEG from juvenile and adult zebra finches for several nights. Whereas adults spent more time in SWS and REM sleep, juveniles spent more time in intermediate sleep (IS). The amount of IS was significantly larger in male juveniles engaged in vocal learning compared to female juveniles, which suggests that IS could be important for vocal learning. In addition, we observed that functional connectivity increased rapidly during maturation of young juveniles, and was stable or declined at older ages. Synchronous activity during sleep was larger for recording sites in the left hemisphere for both juveniles and adults, and generally intra-hemispheric synchrony was larger than inter-hemispheric synchrony during sleep. A graph theory analysis revealed that in adults, highly correlated EEG activity tended to be distributed across fewer networks that were spread across a wider area of the brain, whereas in juveniles, highly correlated EEG activity was distributed across more numerous, albeit smaller, networks in the brain. Overall, our results reveal that significant changes occur in the neural signatures of sleep during maturation in an avian brain.
© 2023. The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures


Similar articles
-
Mammalian-like features of sleep structure in zebra finches.Proc Natl Acad Sci U S A. 2008 Jul 1;105(26):9081-6. doi: 10.1073/pnas.0703452105. Epub 2008 Jun 25. Proc Natl Acad Sci U S A. 2008. PMID: 18579776 Free PMC article.
-
Evolution of slow-wave sleep and palliopallial connectivity in mammals and birds: a hypothesis.Brain Res Bull. 2006 Mar 15;69(1):20-9. doi: 10.1016/j.brainresbull.2005.11.002. Epub 2005 Dec 1. Brain Res Bull. 2006. PMID: 16464681 Review.
-
Budgerigars have complex sleep structure similar to that of mammals.PLoS Biol. 2020 Nov 17;18(11):e3000929. doi: 10.1371/journal.pbio.3000929. eCollection 2020 Nov. PLoS Biol. 2020. PMID: 33201883 Free PMC article.
-
Bird-like propagating brain activity in anesthetized Nile crocodiles.Sleep. 2018 Aug 1;41(8). doi: 10.1093/sleep/zsy105. Sleep. 2018. PMID: 29955880
-
Avian sleep homeostasis: convergent evolution of complex brains, cognition and sleep functions in mammals and birds.Neurosci Biobehav Rev. 2009 Mar;33(3):253-70. doi: 10.1016/j.neubiorev.2008.08.010. Epub 2008 Aug 22. Neurosci Biobehav Rev. 2009. PMID: 18789355 Review.
Cited by
-
Local sleep in songbirds: different simultaneous sleep states across the avian pallium.J Sleep Res. 2025 Jun;34(3):e14344. doi: 10.1111/jsr.14344. Epub 2024 Oct 19. J Sleep Res. 2025. PMID: 39425588 Free PMC article.
-
Song-like activation of syringeal and respiratory muscles during sleep in canaries.J Comp Physiol A Neuroethol Sens Neural Behav Physiol. 2025 Mar;211(2):135-148. doi: 10.1007/s00359-024-01720-7. Epub 2024 Oct 15. J Comp Physiol A Neuroethol Sens Neural Behav Physiol. 2025. PMID: 39404890
-
Harnessing Simple Animal Models to Decode Sleep Mysteries.Mol Biotechnol. 2024 Nov 23. doi: 10.1007/s12033-024-01318-z. Online ahead of print. Mol Biotechnol. 2024. PMID: 39579174 Review.
References
-
- Klein M, Michel F, Jouvet M. Etude Polygraphique du Sommeil ches les Oiseaux. C. R. Seances Soc. Biol. Fil. 1964;158:99–103. - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources

