Mitochondrial N-formyl methionine peptides contribute to exaggerated neutrophil activation in patients with COVID-19
- PMID: 37248708
- PMCID: PMC10231045
- DOI: 10.1080/21505594.2023.2218077
Mitochondrial N-formyl methionine peptides contribute to exaggerated neutrophil activation in patients with COVID-19
Abstract
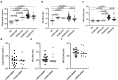
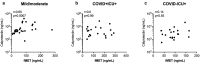
Neutrophil dysregulation is well established in COVID-19. However, factors contributing to neutrophil activation in COVID-19 are not clear. We assessed if N-formyl methionine (fMet) contributes to neutrophil activation in COVID-19. Elevated levels of calprotectin, neutrophil extracellular traps (NETs) and fMet were observed in COVID-19 patients (n = 68), particularly in critically ill patients, as compared to HC (n = 19, p < 0.0001). Of note, the levels of NETs were higher in ICU patients with COVID-19 than in ICU patients without COVID-19 (p < 0.05), suggesting a prominent contribution of NETs in COVID-19. Additionally, plasma from COVID-19 patients with mild and moderate/severe symptoms induced in vitro neutrophil activation through fMet/FPR1 (formyl peptide receptor-1) dependent mechanisms (p < 0.0001). fMet levels correlated with calprotectin levels validating fMet-mediated neutrophil activation in COVID-19 patients (r = 0.60, p = 0.0007). Our data indicate that fMet is an important factor contributing to neutrophil activation in COVID-19 disease and may represent a potential target for therapeutic intervention.
Keywords: COVID-19; N-formyl methionine peptide; clinical biomarkers; mitochondrial peptides; neutrophils; systemic inflammation.
Conflict of interest statement
No potential conflict of interest was reported by the authors.
Figures



References
-
- Reusch N, De Domenico E, Bonaguro L, et al. Neutrophils in COVID-19, Front. Immunol. 2021. [accessed August 19, 2022];12. https://www.frontiersin.org/articles/10.3389/fimmu.2021.652470 - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
