Blood tau-PT217 contributes to the anesthesia/surgery-induced delirium-like behavior in aged mice
- PMID: 37249148
- PMCID: PMC10524579
- DOI: 10.1002/alz.13118
Blood tau-PT217 contributes to the anesthesia/surgery-induced delirium-like behavior in aged mice
Abstract
Introduction: Blood phosphorylated tau at threonine 217 (tau-PT217) is a newly established biomarker for Alzheimer's disease and postoperative delirium in patients. However, the mechanisms and consequences of acute changes in blood tau-PT217 remain largely unknown.
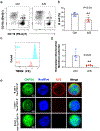
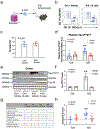
Methods: We investigated the effects of anesthesia/surgery on blood tau-PT217 in aged mice, and evaluated the associated changes in B cell populations, neuronal excitability in anterior cingulate cortex, and delirium-like behavior using positron emission tomography imaging, nanoneedle technology, flow cytometry, electrophysiology, and behavioral tests.
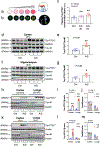
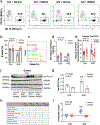
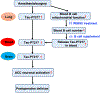
Results: Anesthesia/surgery induced acute increases in blood tau-PT217 via enhanced generation in the lungs and release from B cells. Tau-PT217 might cross the blood-brain barrier, increasing neuronal excitability and inducing delirium-like behavior. B cell transfer and WS635, a mitochondrial function enhancer, mitigated the anesthesia/surgery-induced changes.
Discussion: Acute increases in blood tau-PT217 may contribute to brain dysfunction and postoperative delirium. Targeting B cells or mitochondrial function may have therapeutic potential for preventing or treating these conditions.
Keywords: anesthesia; delirium; phosphorylated tau at threonine 217; surgery; tau; tau phosphorylation.
© 2023 the Alzheimer's Association.
Conflict of interest statement
Figures

References
-
- Vutskits L, Xie Z. Lasting impact of general anaesthesia on the brain: mechanisms and relevance. Nat Rev Neurosci. 2016;17:705–17. - PubMed
-
- Inouye SK. Delirium in older persons. N Engl J Med. 2006;354:1157–65. - PubMed
-
- Rudolph JL, Jones RN, Rasmussen LS, Silverstein JH, Inouye SK, Marcantonio ER. Independent vascular and cognitive risk factors for postoperative delirium. Am J Med. 2007;120:807–13. - PubMed
-
- Witlox J, Eurelings LS, de Jonghe JF, Kalisvaart KJ, Eikelenboom P, van Gool WA. Delirium in elderly patients and the risk of postdischarge mortality, institutionalization, and dementia: a meta-analysis. JAMA. 2010;304:443–51. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases

