MR-based follow-up after brachytherapy and proton beam therapy in uveal melanoma
- PMID: 37249621
- PMCID: PMC10227806
- DOI: 10.1007/s00234-023-03166-1
MR-based follow-up after brachytherapy and proton beam therapy in uveal melanoma
Abstract
Purpose: MRI is increasingly used in the diagnosis and therapy planning of uveal melanoma (UM). In this prospective cohort study, we assessed the radiological characteristics, in terms of anatomical and functional imaging, of UM after ruthenium-106 plaque brachytherapy or proton beam therapy (PBT) and compared them to conventional ultrasound.
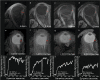
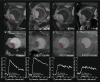
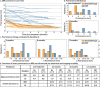
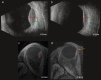
Methods: Twenty-six UM patients were evaluated before and 3, 6 and 12 months after brachytherapy (n = 13) or PBT (n = 13). Tumour prominences were compared between ultrasound and MRI. On diffusion-weighted imaging, the apparent diffusion value (ADC), and on perfusion-weighted imaging (PWI), the time-intensity curves (TIC), relative peak intensity and outflow percentages were determined. Values were compared between treatments and with baseline.
Results: Pre-treatment prominences were comparable between MRI and ultrasound (mean absolute difference 0.51 mm, p = 0.46), but larger differences were observed post-treatment (e.g. 3 months: 0.9 mm (p = 0.02)). Pre-treatment PWI metrics were comparable between treatment groups. After treatment, brachytherapy patients showed favourable changes on PWI (e.g. 67% outflow reduction at 3 months, p < 0.01). After PBT, significant perfusion changes were observed at a later timepoint (e.g. 38% outflow reduction at 6 months, p = 0.01). No consistent ADC changes were observed after either treatment, e.g. a 0.11 × 10-3mm2/s increase 12 months after treatment (p = 0.15).
Conclusion: MR-based follow-up is valuable for PBT-treated patients as favourable perfusion changes, including a reduction in outflow, can be detected before a reduction in size is apparent on ultrasound. For brachytherapy, a follow-up MRI is of less value as already 3 months post-treatment a significant size reduction can be measured on ultrasound.
Keywords: Brachytherapy; Magnetic resonance imaging; Proton beam therapy; Ultrasound; Uveal melanoma.
© 2023. The Author(s).
Conflict of interest statement
The C.J. Gorter Center received research support from Philips Healthcare.
Figures




Similar articles
-
Uveal melanoma: quantitative evaluation of diffusion-weighted MR imaging in the response assessment after proton-beam therapy, long-term follow-up.Radiol Med. 2017 Feb;122(2):131-139. doi: 10.1007/s11547-016-0697-3. Epub 2016 Oct 17. Radiol Med. 2017. PMID: 27752969
-
Neoadjuvant proton beam irradiation vs. adjuvant ruthenium brachytherapy in transscleral resection of uveal melanoma.Graefes Arch Clin Exp Ophthalmol. 2018 Sep;256(9):1767-1775. doi: 10.1007/s00417-018-4032-7. Epub 2018 Jun 16. Graefes Arch Clin Exp Ophthalmol. 2018. PMID: 29907945
-
Quantitative echography in primary uveal melanoma treated by proton beam therapy.Can J Ophthalmol. 2014 Feb;49(1):60-5. doi: 10.1016/j.jcjo.2013.09.007. Can J Ophthalmol. 2014. PMID: 24513359
-
Deterioration of Visual Acuity after Brachytherapy and Proton Therapy of Uveal Melanoma, and Methods of Counteracting This Complication Based on Recent Publications.Medicina (Kaunas). 2023 Jun 12;59(6):1131. doi: 10.3390/medicina59061131. Medicina (Kaunas). 2023. PMID: 37374335 Free PMC article. Review.
-
Uveal melanomas. Conservation treatment.Hematol Oncol Clin North Am. 2001 Apr;15(2):389-402. doi: 10.1016/s0889-8588(05)70219-7. Hematol Oncol Clin North Am. 2001. PMID: 11370500 Review.
Cited by
-
Quantitative Perfusion-Weighted Magnetic Resonance Imaging in Uveal Melanoma.Invest Ophthalmol Vis Sci. 2024 Sep 3;65(11):17. doi: 10.1167/iovs.65.11.17. Invest Ophthalmol Vis Sci. 2024. PMID: 39250118 Free PMC article.
-
Proton Therapy in Uveal Melanoma.Cancers (Basel). 2024 Oct 16;16(20):3497. doi: 10.3390/cancers16203497. Cancers (Basel). 2024. PMID: 39456591 Free PMC article. Review.
-
Clinical outcomes and risk factors for local failure and visual impairment in patients treated with Ru-106 brachytherapy for uveal melanoma.Clin Transl Radiat Oncol. 2025 Feb 24;52:100939. doi: 10.1016/j.ctro.2025.100939. eCollection 2025 May. Clin Transl Radiat Oncol. 2025. PMID: 40110361 Free PMC article.
-
Uncertainties in outcome modelling in radiation oncology.Phys Imaging Radiat Oncol. 2025 May 7;34:100774. doi: 10.1016/j.phro.2025.100774. eCollection 2025 Apr. Phys Imaging Radiat Oncol. 2025. PMID: 40487722 Free PMC article. Review.
-
Estimating uveal melanoma volume with ellipsoid tumour models.Acta Ophthalmol. 2025 Sep;103(6):691-698. doi: 10.1111/aos.17492. Epub 2025 Apr 3. Acta Ophthalmol. 2025. PMID: 40176717 Free PMC article.
References
-
- Dutch Cancer Registry [Accessed on June 10th 2022] [Available from: https://iknl.nl/.
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources

