Nanointerfaces: Concepts and Strategies for Optical and X-ray Spectroscopic Characterization
- PMID: 37249937
- PMCID: PMC10214513
- DOI: 10.1021/acsphyschemau.2c00058
Nanointerfaces: Concepts and Strategies for Optical and X-ray Spectroscopic Characterization
Abstract
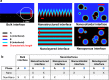
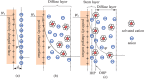
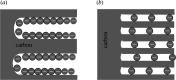
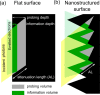
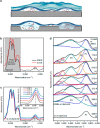
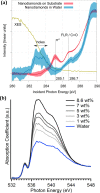
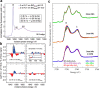
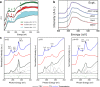
Interfaces at the nanoscale, also called nanointerfaces, play a fundamental role in physics and chemistry. Probing the chemical and electronic environment at nanointerfaces is essential in order to elucidate chemical processes relevant for applications in a variety of fields. Many spectroscopic techniques have been applied for this purpose, although some approaches are more appropriate than others depending on the type of the nanointerface and the physical properties of the different phases. In this Perspective, we introduce the major concepts to be considered when characterizing nanointerfaces. In particular, the interplay between the characteristic length of the nanointerfaces, and the probing and information depths of different spectroscopy techniques is discussed. Differences between nano- and bulk interfaces are explained and illustrated with chosen examples from optical and X-ray spectroscopies, focusing on solid-liquid nanointerfaces. We hope that this Perspective will help to prepare spectroscopic characterization of nanointerfaces and stimulate interest in the development of new spectroscopic techniques adapted to the nanointerfaces.
© 2023 The Authors. Published by American Chemical Society.
Conflict of interest statement
The authors declare no competing financial interest.
Figures




References
-
- Barry E.; Burns R.; Chen W.; De Hoe G. X.; De Oca J. M. M.; De Pablo J. J.; Dombrowski J.; Elam J. W.; Felts A. M.; Galli G.; Hack J.; He Q.; He X.; Hoenig E.; Iscen A.; Kash B.; Kung H. H.; Lewis N. H. C.; Liu C.; Ma X.; Mane A.; Martinson A. B. F.; Mulfort K. L.; Murphy J.; Mølhave K.; Nealey P.; Qiao Y.; Rozyyev V.; Schatz G. C.; Sibener S. J.; Talapin D.; Tiede D. M.; Tirrell M. V.; Tokmakoff A.; Voth G. A.; Wang Z.; Ye Z.; Yesibolati M.; Zaluzec N. J.; Darling S. B. Advanced Materials for Energy-Water Systems: The Central Role of Water/Solid Interfaces in Adsorption, Reactivity, and Transport. Chem. Rev. 2021, 121 (15), 9450–9501. 10.1021/acs.chemrev.1c00069. - DOI - PubMed
Publication types
LinkOut - more resources
Full Text Sources
