Novel Eco-friendly, One-Pot Method for the Synthesis of Kynurenic Acid Ethyl Esters
- PMID: 37251176
- PMCID: PMC10210203
- DOI: 10.1021/acsomega.3c01170
Novel Eco-friendly, One-Pot Method for the Synthesis of Kynurenic Acid Ethyl Esters
Abstract
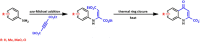
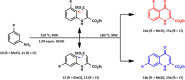
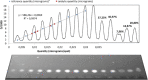
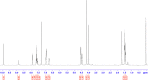
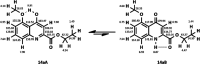
The synthesis of kynurenic acid derivatives with potential biological effect was investigated and optimized for one-batch, two-step microwave-assisted reactions. Utilizing both chemically and biologically representative non-, methyl-, methoxy-, and chlorosubstituted aniline derivatives, in catalyst-free conditions, syntheses of seven kynurenic acid derivatives were achieved in a time frame of 2-3.5 h. In place of halogenated reaction media, tuneable green solvents were introduced for each analogue. The potential of green solvent mixtures to replace traditional solvents and to alter the regioisomeric ratio regarding the Conrad-Limpach method was highlighted. The advantages of the fast, eco-friendly, inexpensive analytic technique of TLC densitometry were emphasized for reaction monitoring and conversion determination in comparison to quantitative NMR. Moreover, the developed 2-3.5 h syntheses were scaled-up to achieve gram-scale products of KYNA derivatives, without altering the reaction time in the halogenated solvent DCB and more importantly in its green substitutes.
© 2023 The Authors. Published by American Chemical Society.
Conflict of interest statement
The authors declare no competing financial interest.
Figures
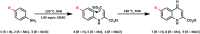


References
-
- Anastas P. T.; Warner J. C.. Green Chemistry: Theory and Practice; Oxford University Press: New York,, 1998.
-
- Byrne F. P.; Jin S.; Paggiola G.; Petchey T. H. M.; Clark J. H.; Farmer T. J.; Hunt A. J.; Robert McElroy C.; Sherwood J. Tools and Techniques for Solvent Selection: Green Solvent Selection Guides. Sustain Chem Process 2016, 4, 7 10.1186/s40508-016-0051-z. - DOI
-
- Capello C.; Fischer U.; Hungerbühler K. What Is a Green Solvent? A Comprehensive Framework for the Environmental Assessment of Solvents. Green Chem. 2007, 9, 927. 10.1039/b617536h. - DOI
LinkOut - more resources
Full Text Sources

