Orchestration of synaptic functions by WAVE regulatory complex-mediated actin reorganization
- PMID: 37258575
- PMCID: PMC10318009
- DOI: 10.1038/s12276-023-01004-1
Orchestration of synaptic functions by WAVE regulatory complex-mediated actin reorganization
Abstract
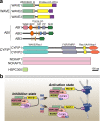
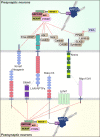
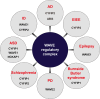
The WAVE regulatory complex (WRC), composed of five components-Cyfip1/Sra1, WAVE/Scar, Abi, Nap1/Nckap1, and Brk1/HSPC300-is essential for proper actin cytoskeletal dynamics and remodeling in eukaryotic cells, likely by matching various patterned signals to Arp2/3-mediated actin nucleation. Accumulating evidence from recent studies has revealed diverse functions of the WRC in neurons, demonstrating its crucial role in dictating the assembly of molecular complexes for the patterning of various trans-synaptic signals. In this review, we discuss recent exciting findings on the physiological role of the WRC in regulating synaptic properties and highlight the involvement of WRC dysfunction in various brain disorders.
© 2023. The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures
Similar articles
-
Nudel is crucial for the WAVE complex assembly in vivo by selectively promoting subcomplex stability and formation through direct interactions.Cell Res. 2012 Aug;22(8):1270-84. doi: 10.1038/cr.2012.47. Epub 2012 Mar 27. Cell Res. 2012. PMID: 22453242 Free PMC article.
-
Differential functions of WAVE regulatory complex subunits in the regulation of actin-driven processes.Eur J Cell Biol. 2017 Dec;96(8):715-727. doi: 10.1016/j.ejcb.2017.08.003. Epub 2017 Sep 4. Eur J Cell Biol. 2017. PMID: 28889942
-
Scar/WAVE drives actin protrusions independently of its VCA domain using proline-rich domains.Curr Biol. 2024 Oct 7;34(19):4436-4451.e9. doi: 10.1016/j.cub.2024.08.013. Epub 2024 Sep 26. Curr Biol. 2024. PMID: 39332399
-
Targeting the WASF3 complex to suppress metastasis.Pharmacol Res. 2022 Aug;182:106302. doi: 10.1016/j.phrs.2022.106302. Epub 2022 Jun 9. Pharmacol Res. 2022. PMID: 35691539 Free PMC article. Review.
-
Regulation of actin assembly by SCAR/WAVE proteins.Biochem Soc Trans. 2005 Dec;33(Pt 6):1243-6. doi: 10.1042/BST0331243. Biochem Soc Trans. 2005. PMID: 16246088 Review.
Cited by
-
SKAP2-A Molecule at the Crossroads for Integrin Signalling and Immune Cell Migration and Function.Biomedicines. 2023 Oct 14;11(10):2788. doi: 10.3390/biomedicines11102788. Biomedicines. 2023. PMID: 37893161 Free PMC article. Review.
-
Neither alpha-synuclein fibril strain nor host murine genotype influences seeding efficacy.NPJ Parkinsons Dis. 2024 May 21;10(1):105. doi: 10.1038/s41531-024-00679-1. NPJ Parkinsons Dis. 2024. PMID: 38773124 Free PMC article.
-
Disulfidptosis decoded: a journey through cell death mysteries, regulatory networks, disease paradigms and future directions.Biomark Res. 2024 Apr 29;12(1):45. doi: 10.1186/s40364-024-00593-x. Biomark Res. 2024. PMID: 38685115 Free PMC article. Review.
-
The impact of rare genetic variants on Alzheimer disease.Nat Rev Neurol. 2025 Mar;21(3):127-139. doi: 10.1038/s41582-025-01062-1. Epub 2025 Feb 4. Nat Rev Neurol. 2025. PMID: 39905212 Review.
-
Impact of Genetic Variants Associated with Neurodevelopmental Disorders on the WAVE Regulatory Complex.J Chem Inf Model. 2025 Jul 28;65(14):7399-7405. doi: 10.1021/acs.jcim.5c01162. Epub 2025 Jul 9. J Chem Inf Model. 2025. PMID: 40633098 Free PMC article.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials

