Added Value of Scintillating Element in Cerenkov-Induced Photodynamic Therapy
- PMID: 37259295
- PMCID: PMC9963809
- DOI: 10.3390/ph16020143
Added Value of Scintillating Element in Cerenkov-Induced Photodynamic Therapy
Abstract
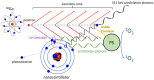
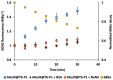
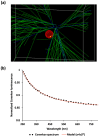
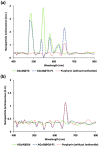
Cerenkov-induced photodynamic therapy (CR-PDT) with the use of Gallium-68 (68Ga) as an unsealed radioactive source has been proposed as an alternative strategy to X-ray-induced photodynamic therapy (X-PDT). This new strategy still aims to produce a photodynamic effect with the use of nanoparticles, namely, AGuIX. Recently, we replaced Gd from the AGuIX@ platform with Terbium (Tb) as a nanoscintillator and added 5-(4-carboxyphenyl succinimide ester)-10,15,20-triphenylporphyrin (P1) as a photosensitizer (referred to as AGuIX@Tb-P1). Although Cerenkov luminescence from 68Ga positrons is involved in nanoscintillator and photosensitizer activation, the cytotoxic effect obtained by PDT remains controversial. Herein, we tested whether free 68Ga could substitute X-rays of X-PDT to obtain a cytotoxic phototherapeutic effect. Results were compared with those obtained with AGuIX@Gd-P1 nanoparticles. We showed, by Monte Carlo simulations, the contribution of Tb scintillation in P1 activation by an energy transfer between Tb and P1 after Cerenkov radiation, compared to the Gd-based nanoparticles. We confirmed the involvement of the type II PDT reaction during 68Ga-mediated Cerenkov luminescence, id est, the transfer of photon to AGuIX@Tb-P1 which, in turn, generated P1-mediated singlet oxygen. The effect of 68Ga on cell survival was studied by clonogenic assays using human glioblastoma U-251 MG cells. Exposure of pre-treated cells with AGuIX@Tb-P1 to 68Ga resulted in the decrease in cell clone formation, unlike AGuIX@Gd-P1. We conclude that CR-PDT could be an alternative of X-PDT.
Keywords: AGuIX; Cerenkov radiation; Gadolinium; Monte Carlo simulations; Terbium; glioblastoma; photodynamic therapy; singlet oxygen.
Conflict of interest statement
The AGuIX design is protected by patent WO2011135101 (F.L. and O.T.). Elsewhere, the authors report no conflict of interest in this work.
Figures

Similar articles
-
Terbium-Based AGuIX-Design Nanoparticle to Mediate X-ray-Induced Photodynamic Therapy.Pharmaceuticals (Basel). 2021 Apr 22;14(5):396. doi: 10.3390/ph14050396. Pharmaceuticals (Basel). 2021. PMID: 33922073 Free PMC article.
-
Lanthanides-Based Nanoparticles Conjugated with Rose Bengal for FRET-Mediated X-Ray-Induced PDT.Pharmaceuticals (Basel). 2025 May 1;18(5):672. doi: 10.3390/ph18050672. Pharmaceuticals (Basel). 2025. PMID: 40430491 Free PMC article.
-
Energy Transfer between AGuIX Nanoparticles and Photofrin under Light or X-ray Excitation for PDT Applications.Pharmaceuticals (Basel). 2024 Aug 5;17(8):1033. doi: 10.3390/ph17081033. Pharmaceuticals (Basel). 2024. PMID: 39204138 Free PMC article.
-
Nanoparticles to mediate X-ray-induced photodynamic therapy and Cherenkov radiation photodynamic therapy.Wiley Interdiscip Rev Nanomed Nanobiotechnol. 2019 Mar;11(2):e1541. doi: 10.1002/wnan.1541. Epub 2018 Jul 31. Wiley Interdiscip Rev Nanomed Nanobiotechnol. 2019. PMID: 30063116 Free PMC article. Review.
-
Nanoparticles for Cerenkov and Radioluminescent Light Enhancement for Imaging and Radiotherapy.Nanomaterials (Basel). 2020 Sep 7;10(9):1771. doi: 10.3390/nano10091771. Nanomaterials (Basel). 2020. PMID: 32906838 Free PMC article. Review.
Cited by
-
Investigation of Photodynamic Therapy Promoted by Cherenkov Light Activated Photosensitizers-New Aspects and Revelations.Pharmaceutics. 2024 Apr 13;16(4):534. doi: 10.3390/pharmaceutics16040534. Pharmaceutics. 2024. PMID: 38675195 Free PMC article.
-
Non-targeted effects of radiation therapy for glioblastoma.Heliyon. 2024 May 9;10(10):e30813. doi: 10.1016/j.heliyon.2024.e30813. eCollection 2024 May 30. Heliyon. 2024. PMID: 38778925 Free PMC article. Review.
-
Novel Drug Delivery Particles Can Provide Dual Effects on Cancer "Theranostics" in Boron Neutron Capture Therapy.Cells. 2025 Jan 6;14(1):60. doi: 10.3390/cells14010060. Cells. 2025. PMID: 39791761 Free PMC article.
References
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

