What bothers severe asthma patients most? A paired patient-clinician study across seven European countries
- PMID: 37260457
- PMCID: PMC10227631
- DOI: 10.1183/23120541.00717-2022
What bothers severe asthma patients most? A paired patient-clinician study across seven European countries
Abstract
Introduction: Severe asthma is a complex, multidimensional disease. Optimal treatment, adherence and outcomes require shared decision-making, rooted in mutual understanding between patient and clinician. This study used a novel, patient-centred approach to examine the most bothersome aspects of severe asthma to patients, as seen from both perspectives in asthma registries.
Methods: Across seven countries, 126 patients with severe asthma completed an open-ended survey regarding most the bothersome aspect(s) of their asthma. Patients' responses were linked with their treating clinician who also completed a free-text survey about each patient's most bothersome aspect(s). Responses were coded using content analysis, and patient and clinician responses were compared. Finally, asthma registries that are part of the SHARP (Severe Heterogeneous Asthma Research collaboration, Patient-centred) Clinical Research Collaboration were examined to see the extent to which they reflected the most bothersome aspects reported by patients.
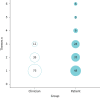
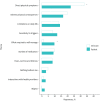
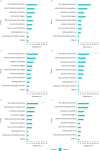
Results: 88 codes and 10 themes were identified. Clinicians were more focused on direct physical symptoms and were less focused on "holistic" aspects such as the effort required to self-manage the disease. Clinicians accurately identified a most bothersome symptom for 29% of patients. Agreement was particularly low with younger patients and those using oral corticosteroids infrequently. In asthma registries, patient aspects were predominantly represented in questionnaires.
Conclusions: Results demonstrated different perspectives and priorities between patients and clinicians, with clinicians more focused on physical aspects. These differences must be considered when treating individual patients, and within multidisciplinary treatment teams. The use of questionnaires that include multifaceted aspects of disease may result in improved asthma research.
Copyright ©The authors 2023.
Conflict of interest statement
Conflict of interest: B. Ainsworth is a member of the UK Taskforce for Lung Health, has received honoraria for educational talks from AstraZeneca, and sits on advisory boards for the Medito Foundation and earGym. Conflict of interest: C. Coleman is an employee of the European Lung Foundation. Conflict of interest: M. Hyland has received grant income from Teva, GSK and AstraZeneca. Conflict of interest: L. Heaney has received grant funding, participated in advisory boards and given lectures at meetings supported by Amgen, AstraZeneca, Boehringer Ingelheim, Chiesi, Circassia, Hoffmann la Roche, GlaxoSmithKline, Novartis, Theravance, Evelo Biosciences, Sanofi and Teva; he has received grants from MedImmune, Novartis UK, Roche/Genentech Inc., GlaxoSmithKline, Amgen, Genentech/Hoffman la Roche, AstraZeneca, MedImmune, Aerocrine and Vitalograph; he has received sponsorship for attending international scientific meetings from AstraZeneca, Boehringer Ingelheim, Chiesi, GSK and Napp Pharmaceuticals; he has also taken part in asthma clinical trials sponsored by AstraZeneca, Boehringer Ingelheim, Hoffmann la Roche and GlaxoSmithKline for which his institution received remuneration; he is the Academic Lead for the Medical Research Council Stratified Medicine UK Consortium in Severe Asthma, which involves industrial partnerships with a number of pharmaceutical companies including Amgen, AstraZeneca, Boehringer Ingelheim, GlaxoSmithKline, Hoffmann la Roche, and Janssen. Conflict of interest: V. Sedlák has received honoraria for educational talks and advisory boards from AstraZeneca, GSK, Novartis, TEVA, Chiesi and Boehringer Ingelheim. Conflict of interest: N. Edelbaher has received honoraria for educational talks and advisory boards from AstraZeneca, Sanofi, Novartis, GSK, Amgen, Boherringer Ingelheim, Chiesi and Berlin Chemie. Conflict of interest: A. ten Brinke has received honoraria for educational talks and advisory board from AstraZeneca, GSK, Novartis, Sanofi and TEVA, and grant income from AstraZeneca, GSK and TEVA. Conflict of interest: C. Loureiro has received honoraria for educational talks and advisory boards from AstraZeneca, GSK, Jaba-Recordati, Novartis, Sanofi and TEVA. Conflict of interest: R. Djukanovic has received honoraria for education talks and advisory board from AZ, GSK, Teva, Novartis and Sanofi. Conflict of interest: N. Kwon is a GSK employee and held stocks in GSK at the time of manuscript writing. Conflict of interest: All other authors have no competing interests.
Figures
References
-
- World Allergy Organization . The Management of Severe Asthma: Economic Analysis of the Cost of Treatments for Severe Asthma. 2005. www.worldallergy.org/educational_programs/world_allergy_forum/anaheim200.... Date last accessed: June 2018.
Associated data
LinkOut - more resources
Full Text Sources
