Antiangiogenic Potential of Troxerutin and Chitosan Loaded Troxerutin on Chorioallantoic Membrane Model
- PMID: 37260851
- PMCID: PMC10229255
- DOI: 10.1155/2023/5956154
Antiangiogenic Potential of Troxerutin and Chitosan Loaded Troxerutin on Chorioallantoic Membrane Model
Abstract
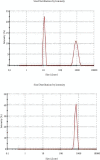
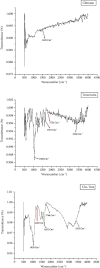
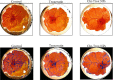
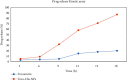
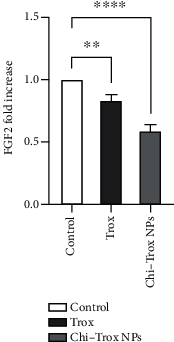
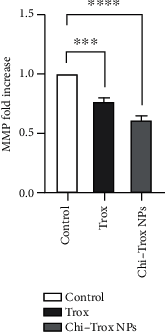
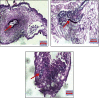
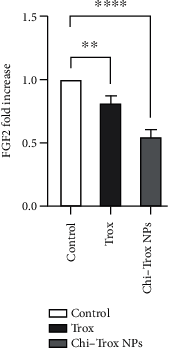
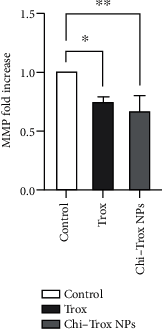
Angiogenesis is crucial to the development of cancer because it allows the transport of oxygen, nutrients, and growth factors as well as the spread of tumors to distant organs. Inhibitors of angiogenesis prevent the formation of blood vessels that allow tumor cells to shrink, rather than promote tumor growth. Chitosan acts as a carrier for many drugs, since the compound has various properties such as biodegradable, less toxicity, more stable, simple, easy to prepare, and biocompatible. The aim of the current study was to evaluate the efficacy of chitosan nanoparticles encapsulated with troxerutin (Chi-Trox NPs) against angiogenesis and cancer in ova chick chorioallantoic membrane (CAM) model. Chi-Trox NPs were synthesized using a nanoprecipitation method and were characterized by various analyses. 24 hours' fertilized eggs (6 eggs/group) were treated with native Trox and Chi-Trox NPs for 5 days. The antiangiogenic activity was evaluated by morphometric, histopathological, immunohistochemical (CD104 and vimentin), and mRNA expression of MMP and FGF2 using RT-PCR. The anticancer activity was evaluated by histopathological, immunohistochmical (CD44), and mRNA expression of FGF2 and MMP. The synthesized chitosan NPs were successfully encapsulated with troxerutin, and the loading efficiency of chitosan NPs was found to be 86.4 ± 0.12% and 13.2 ± 0.16% respectively. Morphometric analysis of Chi-Trox NPs showed a considerable decrease in the number of blood vessels compared with control and native Trox. The histopathological observation of CAM confirmed that Chi-Trox NPs induce a significant reduction in inflammatory cells and the thickness of blood capillaries compared to control and native Trox. The immunohistochemical evaluation of CAM revealed Chi-Trox decreased CD104, vimentin and CD44 protein levels were compared with control and native Trox. Furthermore, the mRNA expression levels of FGF2 and MMP were significantly downregulated compared to their native forms. From the obtained results, Chi-Trox NPs possess significant inhibition of angiogenesis and can be used as therapeutic agents for cancer in the future.
Copyright © 2023 Gowtham Kumar Subbaraj et al.
Conflict of interest statement
The authors declare that they have no competing interests.
Figures






Similar articles
-
The Protective Role of Troxerutin (Trox) in Counteracting Anaplastic Thyroid Carcinoma (ATC) Progression.Biomedicines. 2024 Aug 5;12(8):1755. doi: 10.3390/biomedicines12081755. Biomedicines. 2024. PMID: 39200219 Free PMC article.
-
Ursolic acid-loaded chitosan nanoparticles induce potent anti-angiogenesis in tumor.Appl Microbiol Biotechnol. 2016 Aug;100(15):6643-6652. doi: 10.1007/s00253-016-7360-8. Epub 2016 Feb 17. Appl Microbiol Biotechnol. 2016. PMID: 26883344
-
Anti-angiogenic effect of nano-formulated water soluble kaempferol and combretastatin in an in vivo chick chorioallantoic membrane model and HUVEC cells.Biomed Pharmacother. 2023 Jul;163:114820. doi: 10.1016/j.biopha.2023.114820. Epub 2023 May 2. Biomed Pharmacother. 2023. PMID: 37141736
-
Treatment of oxidative stress-induced pain and inflammation with dexketoprofen trometamol loaded different molecular weight chitosan nanoparticles: Formulation, characterization and anti-inflammatory activity by using in vivo HET-CAM assay.Microvasc Res. 2020 Mar;128:103961. doi: 10.1016/j.mvr.2019.103961. Epub 2019 Nov 20. Microvasc Res. 2020. PMID: 31758946
-
Understanding the Factors Influencing Chitosan-Based Nanoparticles-Protein Corona Interaction and Drug Delivery Applications.Molecules. 2020 Oct 16;25(20):4758. doi: 10.3390/molecules25204758. Molecules. 2020. PMID: 33081296 Free PMC article. Review.
Cited by
-
Integrating Blood Biomarkers and Marine Brown Algae-Derived Inhibitors in Parkinson's Disease: A Multi-scale Approach from Interactomics to Quantum Mechanics.Mol Biotechnol. 2024 Sep 3. doi: 10.1007/s12033-024-01262-y. Online ahead of print. Mol Biotechnol. 2024. PMID: 39225961
-
Troxerutin suppress inflammation response and oxidative stress in jellyfish dermatitis by activating Nrf2/HO-1 signaling pathway.Front Immunol. 2024 May 8;15:1369849. doi: 10.3389/fimmu.2024.1369849. eCollection 2024. Front Immunol. 2024. PMID: 38779681 Free PMC article.
-
Effect of bovine milk-derived peptide on SNAP-25 of the neurotransmitter system in treating the sialorrhoea in chronic neurological diseases.Food Sci Biotechnol. 2025 Mar 29;34(11):2601-2610. doi: 10.1007/s10068-025-01872-5. eCollection 2025 Jul. Food Sci Biotechnol. 2025. PMID: 40492044
-
The Protective Role of Troxerutin (Trox) in Counteracting Anaplastic Thyroid Carcinoma (ATC) Progression.Biomedicines. 2024 Aug 5;12(8):1755. doi: 10.3390/biomedicines12081755. Biomedicines. 2024. PMID: 39200219 Free PMC article.
-
Metalloproteinases in Restorative Dentistry: An In Silico Study toward an Ideal Animal Model.Biomedicines. 2023 Nov 14;11(11):3042. doi: 10.3390/biomedicines11113042. Biomedicines. 2023. PMID: 38002041 Free PMC article.
References
-
- Abdolmaleki Z., Arab H. A., Amanpour S., Muhammadnejad S. Anti-angiogenic effects of ethanolic extract of Artemisia sieberi compared to its active substance, artemisinin. Revista Brasileira de Farmacognosia . 2016;26(3):326–333. doi: 10.1016/j.bjp.2015.11.008. - DOI
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous

