Immunotherapy in oncology and the kidneys: a clinical review of the evaluation and management of kidney immune-related adverse events
- PMID: 37261008
- PMCID: PMC10229281
- DOI: 10.1093/ckj/sfad014
Immunotherapy in oncology and the kidneys: a clinical review of the evaluation and management of kidney immune-related adverse events
Abstract
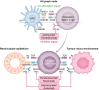
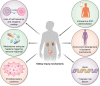
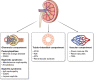
Immune checkpoint inhibitors (ICI) are now widely used in the treatment of many cancers, and currently represent the standard of care for multiple malignancies. These agents enhance the T cell immune response to target cancer tissues, and have demonstrated considerable benefits for cancer outcomes. However, despite these improved outcomes, there are important kidney immune-related adverse events (iRAEs) associated with ICI. Acute tubulo-interstitial nephritis remains the most frequent kidney iRAE, however glomerular lesions and electrolytes disturbances are increasingly being recognized and reported. In this review, we summarize clinical features and identify risk factors for kidney iRAEs, and discuss the current understanding of pathophysiologic mechanisms. We highlight the evidence basis for guideline-recommended management of ICI-related kidney injury as well as gaps in current knowledge. We advocate for judicious use of kidney biopsy to identify ICI-associated kidney injury, and early use of corticosteroid treatment where appropriate. Selected patients may also be candidates for re-challenge with ICI therapy after a kidney iRAE, in view of current data on recurrent rates of kidney injury. Risk of benefits of re-challenge must be considered on an individual considering patient preferences and prognosis. Lastly, we review current knowledge of ICI use in the setting of patients with end-stage kidney disease, including kidney transplant recipients and those receiving dialysis, which suggest that these patients should not be summarily excluded from the potential benefits of these cancer therapies.
Keywords: AKI; cancer; glomerulonephritis; immune checkpoint inhibitors; nephrotoxicity.
© The Author(s) 2023. Published by Oxford University Press on behalf of the ERA.
Conflict of interest statement
None declared.
Figures
Similar articles
-
Immune checkpoint inhibitor related nephrotoxicity: Advances in clinicopathologic features, noninvasive approaches, and therapeutic strategy and rechallenge.Front Nephrol. 2022 Oct 19;2:1017921. doi: 10.3389/fneph.2022.1017921. eCollection 2022. Front Nephrol. 2022. PMID: 37674988 Free PMC article. Review.
-
Immune checkpoint inhibitor-associated nephritis-treatment standard.Nephrol Dial Transplant. 2024 Oct 30;39(11):1785-1798. doi: 10.1093/ndt/gfae184. Nephrol Dial Transplant. 2024. PMID: 39138117 Review.
-
Histological diagnosis of immune checkpoint inhibitor induced acute renal injury in patients with metastatic melanoma: a retrospective case series report.BMC Nephrol. 2020 Sep 7;21(1):391. doi: 10.1186/s12882-020-02044-9. BMC Nephrol. 2020. PMID: 32894101 Free PMC article.
-
Real-World Clinical and Economic Outcomes in Selected Immune-Related Adverse Events Among Patients with Cancer Receiving Immune Checkpoint Inhibitors.Oncologist. 2021 Nov;26(11):e2002-e2012. doi: 10.1002/onco.13918. Epub 2021 Aug 24. Oncologist. 2021. PMID: 34327774 Free PMC article.
-
Risk factors associated with immune checkpoint inhibitor-induced acute kidney injury compared with other immune-related adverse events: a case-control study.Clin Kidney J. 2022 Apr 28;15(10):1881-1887. doi: 10.1093/ckj/sfac109. eCollection 2022 Oct. Clin Kidney J. 2022. PMID: 36158153 Free PMC article.
Cited by
-
Clinical features associated with immune checkpoint inhibitor nephritis: a single-center clinical case series.Cancer Immunol Immunother. 2024 Aug 6;73(10):200. doi: 10.1007/s00262-024-03775-6. Cancer Immunol Immunother. 2024. PMID: 39105812 Free PMC article.
-
Higher Risk of Proteinuria with Atezolizumab plus Bevacizumab than Lenvatinib in First-Line Systemic Treatment for Hepatocellular Carcinoma.Liver Cancer. 2024 Oct 16;14(2):180-192. doi: 10.1159/000541621. eCollection 2025 Apr. Liver Cancer. 2024. PMID: 40255873 Free PMC article.
-
Renal Immune-Related Adverse Event Difficult to Diagnose during Nivolumab Treatment for Hypopharyngeal Carcinoma: Case Report.Case Rep Oncol. 2024 Aug 30;17(1):966-972. doi: 10.1159/000540373. eCollection 2024 Jan-Dec. Case Rep Oncol. 2024. PMID: 39474555 Free PMC article.
-
Pneumocystis pneumonia in stage IIIA lung adenocarcinoma with immune-related acute kidney injury and thoracic radiotherapy: A case report.World J Radiol. 2024 Sep 28;16(9):482-488. doi: 10.4329/wjr.v16.i9.482. World J Radiol. 2024. PMID: 39355392 Free PMC article.
-
Complete response of metastatic papillary renal cell carcinoma with inferior vena cava tumor thrombus to nivolumab plus cabozantinib.IJU Case Rep. 2023 Sep 5;6(6):419-423. doi: 10.1002/iju5.12638. eCollection 2023 Nov. IJU Case Rep. 2023. PMID: 37928303 Free PMC article.
References
-
- Shirali AC, Perazella MA, Gettinger S.. Association of acute interstitial nephritis with programmed cell death 1 inhibitor therapy in lung cancer patients. Am J Kidney Dis 2016;68:287–91. - PubMed
-
- Thajudeen B, Madhrira M, Bracamonte Eet al. . Ipilimumab granulomatous interstitial nephritis. Am J Ther 2015;22:e84–7. - PubMed
Publication types
LinkOut - more resources
Full Text Sources

