Natural history of retinitis pigmentosa based on genotype, vitamin A/E supplementation, and an electroretinogram biomarker
- PMID: 37261916
- PMCID: PMC10445682
- DOI: 10.1172/jci.insight.167546
Natural history of retinitis pigmentosa based on genotype, vitamin A/E supplementation, and an electroretinogram biomarker
Abstract
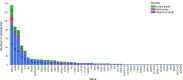
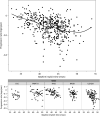
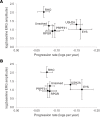
BACKGROUNDA randomized clinical trial from 1984 to 1992 indicated that vitamin A supplementation had a beneficial effect on the progression of retinitis pigmentosa (RP), while vitamin E had an adverse effect.METHODSSequencing of banked DNA samples from that trial provided the opportunity to determine whether certain genotypes responded preferentially to vitamin supplementation.RESULTSThe genetic solution rate was 587 out of 765 (77%) of sequenced samples. Combining genetic solutions with electroretinogram outcomes showed that there were systematic differences in severity and progression seen among different genetic subtypes of RP, extending findings made for USH2A, RHO, RPGR, PRPF31, and EYS. Baseline electroretinogram 30-Hz flicker implicit time was an independent, strong predictor of progression rate. Using additional data and baseline implicit time as a predictor, the deleterious effect of vitamin E was still present. Surprisingly, the effect of vitamin A progression in the cohort as a whole was not detectable, with or without data from subsequent trials. Subgroup analyses are also discussed.CONCLUSIONOverall, genetic subtype and implicit time have significant predictive power for a patient's rate of progression, which is useful prognostically. While vitamin E supplementation should still be avoided, these data do not support a generalized neuroprotective effect of vitamin A for all types of RP.TRIAL REGISTRATIONClinicalTrials.gov NCT00000114, NCT00000116, and NCT00346333.FUNDINGFoundation Fighting Blindness and the National Eye Institute: R01 EY012910, R01 EY031036, R01 EY026904, and P30 EY014104.
Keywords: Ophthalmology; Retinopathy.
Figures