Impact of serum‑free media on the expansion and functionality of CD19.CAR T‑cells
- PMID: 37264971
- PMCID: PMC10249581
- DOI: 10.3892/ijmm.2023.5261
Impact of serum‑free media on the expansion and functionality of CD19.CAR T‑cells
Abstract
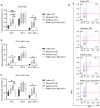
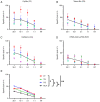
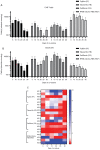
Fetal bovine serum (FBS) or human serum is widely used in the production of chimeric antigen receptor (CAR) T‑cells. In order to overcome a lot‑to‑lot inconsistency, the use of chemically defined medium that is free of animal-components would be highly desirable. The present study compared three serum‑free media [Prime‑XV™ T Cell CDM, Fujifilm™ (FF), LymphoONE™ T‑Cell Expansion Xeno‑Free Medium, Takara Bio™ (TB) and TCM GMP‑Prototype, CellGenix™ (CG)] to the standard CAR T‑cell medium containing FBS (RCF). After 12 days of CD19.CAR T‑cell culture, the expansion, viability, transduction efficiency and phenotype were assessed using flow cytometry. The functionality of CAR T‑cells was evaluated using intracellular staining, a chromium release assay and a long‑term co‑culture assay. Expansion and viability did not differ between the CAR T‑cells generated in serum‑free media compared to the standard FBS‑containing medium. The CG CAR T‑cells had a statistically significant higher frequency of IFNγ+ and IFNγ+TNF‑α+ CAR T‑cells than the CAR T‑cells cultured with FBS (22.5 vs. 7.6%, P=0.0194; 15.3 vs. 6.2%, P=0.0399, respectively) as detected by intracellular cytokine staining. The CAR T‑cells generated with serum‑free media exhibited a higher cytotoxicity than the CAR T‑cells cultured with FBS in the evaluation by chromium release assay [CG vs. RCF (P=0.0182), FF vs. RCF (P=0.0482) and TB vs. RCF (P=0.0482)]. Phenotyping on day 12 of CAR T‑cell production did not reveal a significant difference in the expression of the exhaustion markers, programmed cell death protein 1, lymphocyte‑activation gene 3 and T‑cell immunoglobulin and mucin‑domain containing‑3. The CAR T‑cells cultured in FF had a higher percentage of central memory CAR T‑cells (40.0 vs. 14.3%, P=0.0470) than the CAR T‑cells cultured with FBS, whereas the CAR T‑cells in FF (6.2 vs. 24.2%, P=0.0029) and CG (11.0% vs. 24.2%, P=0.0468) had a lower frequency of naïve CAR T‑cells. On the whole, the present study demonstrates that in general, the functionality and expansion of CAR T cells are maintained in serum‑free media. Given the advantages of freedom from bovine material and consistent quality, serum‑free media hold promise for the future development of the field of GMP manufacturing of CAR T‑cells.
Keywords: chemically defined medium; chimeric antigen receptor T‑cells; fetal bovine serum; serum‑containing medium; serum‑free medium.
Conflict of interest statement
The authors declare that they have no competing interests.
Figures


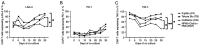
References
-
- Locke FL, Ghobadi A, Jacobson CA, Miklos DB, Lekakis LJ, Oluwole OO, Lin Y, Braunschweig I, Hill BT, Timmerman JM, et al. Long-term safety and activity of axicabtagene ciloleucel in refractory large B-cell lymphoma (ZUMA-1): A single-arm, multicentre, phase 1-2 trial. Lancet Oncol. 2019;20:31–42. doi: 10.1016/S1470-2045(18)30864-7. - DOI - PMC - PubMed
-
- Kochenderfer JN, Dudley ME, Kassim SH, Somerville RP, Carpenter RO, Stetler-Stevenson M, Yang JC, Phan GQ, Hughes MS, Sherry RM, et al. Chemotherapy-refractory diffuse large B-cell lymphoma and indolent B-cell malignancies can be effectively treated with autologous T cells expressing an anti-CD19 chimeric antigen receptor. J Clin Oncol. 2015;33:540–549. doi: 10.1200/JCO.2014.56.2025. - DOI - PMC - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

