Synthesis of spirooxindoles via formal acetylene insertion into a common palladacycle intermediate
- PMID: 37265736
- PMCID: PMC10231318
- DOI: 10.1039/d3sc01072d
Synthesis of spirooxindoles via formal acetylene insertion into a common palladacycle intermediate
Abstract
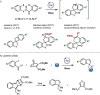
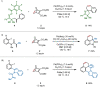
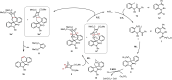
A palladium-catalyzed spirocyclization reaction is reported, which is proposed to arise via insertion of an oxabicycle into a palladacycle, formed from carbocyclization and a C-H functionalization sequence. Mechanistic studies suggest the insertion is diastereoselective and a post-catalytic retro-Diels-Alder step furnishes an alkene, wherein the oxibicycle has served as an acetylene surrogate. Aryl iodides and carbamoyl chlorides were compatible as starting materials under the same reaction conditions, enabling the convergent and complementary synthesis of spirooxindoles, as well as other azacycles. These spirooxindoles allowed further transformations that were previously unaccessible.
This journal is © The Royal Society of Chemistry.
Conflict of interest statement
There are no conflicts to declare.
Figures




Similar articles
-
Palladium-Catalyzed Multicomponent Assembly of (Z)-Alkenylborons via Carbopalladation/Boronation/Retro-Diels-Alder Cascade Reaction.J Org Chem. 2023 Aug 18;88(16):11793-11800. doi: 10.1021/acs.joc.3c01084. Epub 2023 Jul 29. J Org Chem. 2023. PMID: 37515567
-
Palladium-catalyzed C-H glycosylation and retro Diels-Alder tandem reaction via structurally modified norbornadienes (smNBDs).Chem Sci. 2021 Aug 31;12(39):13144-13150. doi: 10.1039/d1sc03569j. eCollection 2021 Oct 13. Chem Sci. 2021. PMID: 34745545 Free PMC article.
-
Palladium-catalyzed synthesis of β-hydroxy compounds via a strained 6,4-palladacycle from directed C-H activation of anilines and C-O insertion of epoxides.Chem Sci. 2020 Jun 22;11(27):7260-7265. doi: 10.1039/d0sc01462a. Chem Sci. 2020. PMID: 34123012 Free PMC article.
-
A novel and efficient synthesis of phenanthrene derivatives via palladium/norbornadiene-catalyzed domino one-pot reaction.Beilstein J Org Chem. 2019 Jan 31;15:291-298. doi: 10.3762/bjoc.15.26. eCollection 2019. Beilstein J Org Chem. 2019. PMID: 30800179 Free PMC article.
-
Palladium(0)-Catalyzed Benzylic C(sp3)-H Functionalization for the Concise Synthesis of Heterocycles and Its Applications.Chem Pharm Bull (Tokyo). 2017;65(5):409-425. doi: 10.1248/cpb.c16-00969. Chem Pharm Bull (Tokyo). 2017. PMID: 28458363 Review.
Cited by
-
Recent Advances in Sequentially Pd-Catalyzed One-Pot Syntheses of Heterocycles.Molecules. 2024 Nov 7;29(22):5265. doi: 10.3390/molecules29225265. Molecules. 2024. PMID: 39598654 Free PMC article. Review.
References
-
-
For select reviews on transition-metal catalyzed C–H functionalization methodologies see:
- Alberico D. Scott M. E. Lautens M. Chem. Rev. 2007;107:174. doi: 10.1021/cr0509760. - DOI - PubMed
- Thansandote P. Lautens M. Chem. - Eur. J. 2009;15:5874. doi: 10.1002/chem.200900281. - DOI - PubMed
- McMurray L. O'Hara F. Gaunt M. J. Chem. Soc. Rev. 2011;40:1885. doi: 10.1039/C1CS15013H. - DOI - PubMed
- Baudoin O. Chem. Soc. Rev. 2011;40:4902. doi: 10.1039/C1CS15058H. - DOI - PubMed
- Liu C. Yuan J. Gao M. Tang S. Li W. Shi R. Lei A. Chem. Rev. 2015;115:12138. doi: 10.1021/cr500431s. - DOI - PubMed
- Hartwig J. F. J. Am. Chem. Soc. 2016;138:2. doi: 10.1021/jacs.5b08707. - DOI - PMC - PubMed
- Xue X.-S. Ji P. Zhou B. Cheng J.-P. Chem. Rev. 2017;117:8622. doi: 10.1021/acs.chemrev.6b00664. - DOI - PubMed
- Font M. Gulías M. Mascareñas J. L. Angew. Chem., Int. Ed. 2022;61:e202112848. doi: 10.1002/anie.202112848. - DOI - PMC - PubMed
- Dutta S. Chatterjee S. Al-Thabaiti S. A. Bawaked S. Mokhtar M. Maiti D. Chem Catal. 2022;2:1046. doi: 10.1016/j.checat.2022.03.027. - DOI
- Lu M.-Z., Goh J., Maraswami M., Jia Z., Tian J.-S. and Loh T.-P., Chem. Rev., 2022, 122, 17479. - PubMed
-
-
-
For select reviews on palladium-catalyzed C–H activation methodologies see:
- Chen X. Engle K. M. Wang D.-H. Yu J.-Q. Angew. Chem., Int. Ed. 2009;48:5094. doi: 10.1002/anie.200806273. - DOI - PMC - PubMed
- Lyons T. W. Sanford M. S. Chem. Rev. 2010;110:1147. doi: 10.1021/cr900184e. - DOI - PMC - PubMed
- Shaikkh T. M. Hong F.-E. J. Organomet. Chem. 2016;801:139. doi: 10.1016/j.jorganchem.2015.10.022. - DOI
- Wang J. Dong G. Chem. Rev. 2019;119:7478. doi: 10.1021/acs.chemrev.9b00079. - DOI - PMC - PubMed
-
-
-
For reports of Pd catalyzed domino-Heck syntheses of spirocycles resulting from direct C–C reductive elimination see:
- Ye J. Shi Z. Sperger T. Yasukawa Y. Kingston C. Schoenebeck F. Lautens M. Nat. Chem. 2017;9:361. doi: 10.1038/nchem.2631. - DOI - PubMed
- Marchese A. D. Durant A. G. Lautens M. Org. Lett. 2022;24:95. doi: 10.1021/acs.orglett.1c03682. - DOI - PubMed
- Xu B. Ji D. Wu L. Zhou L. Liu Y. Zhang Z.-M. Zhang J. Chem. 2022;8:836. doi: 10.1016/j.chempr.2021.12.019. - DOI
-
-
-
For reports of Pd catalyzed domino-Heck syntheses of spirocycles resulting from insertion of an external reagent into a palladacycle intermediate see:
- Zheng H. Zhu Y. Shi Y. Angew. Chem., Int. Ed. 2014;53:11280. doi: 10.1002/anie.201405365. - DOI - PMC - PubMed
- Pérez-Gómez M. García-López J.-A. Angew. Chem., Int. Ed. 2016;55:14389. doi: 10.1002/anie.201607976. - DOI - PubMed
- Yoon H. Lossouarn A. Landau F. Lautens M. Org. Lett. 2016;18:6324. doi: 10.1021/acs.orglett.6b03213. - DOI - PubMed
- Pérez-Gómez M. Hernández-Ponte S. Bautista D. García-López J.-A. Chem. Commun. 2017;53:2842. doi: 10.1039/C7CC00065K. - DOI - PubMed
- Shao C. Wu Z. Ji X. Zhou B. Zhang Y. Chem. Commun. 2017;53:10429. doi: 10.1039/C7CC06196J. - DOI - PubMed
- Yoon H. Rölz M. Landau F. Lautens M. Angew. Chem., Int. Ed. 2017;56:10920. doi: 10.1002/anie.201706325. - DOI - PubMed
- Xiao G. Chen L. Deng G. Liu J. Liang Y. Tetrahedron Lett. 2018;59:1836. doi: 10.1016/j.tetlet.2018.03.086. - DOI
- Ye F. Ge Y. Spannenberg A. Neumann H. Beller M. Nat. Commun. 2020;11:5383. doi: 10.1038/s41467-020-19110-3. - DOI - PMC - PubMed
- Ma J. Liu T.-X. Zhang P. Zhang C. Zhang G. Chem. Commun. 2021;57:49. doi: 10.1039/D0CC07143A. - DOI - PubMed
- Xu Y. Xu W. Chen X. Luo X. Lu H. Zhang M. Yang X. Deng G. Liang Y. Yang Y. Chem. Sci. 2021;12:11756. doi: 10.1039/D1SC03487A. - DOI - PMC - PubMed
-
LinkOut - more resources
Full Text Sources

