Group A Streptococcus induces CD1a-autoreactive T cells and promotes psoriatic inflammation
- PMID: 37267382
- PMCID: PMC7615662
- DOI: 10.1126/sciimmunol.add9232
Group A Streptococcus induces CD1a-autoreactive T cells and promotes psoriatic inflammation
Abstract
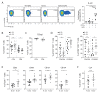
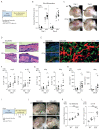
Group A Streptococcus (GAS) infection is associated with multiple clinical sequelae, including different subtypes of psoriasis. Such post-streptococcal disorders have been long known but are largely unexplained. CD1a is expressed at constitutively high levels by Langerhans cells and presents lipid antigens to T cells, but the potential relevance to GAS infection has not been studied. Here, we investigated whether GAS-responsive CD1a-restricted T cells contribute to the pathogenesis of psoriasis. Healthy individuals had high frequencies of circulating and cutaneous GAS-responsive CD4+ and CD8+ T cells with rapid effector functions, including the production of interleukin-22 (IL-22). Human skin and blood single-cell CITE-seq analyses of IL-22-producing T cells showed a type 17 signature with proliferative potential, whereas IFN-γ-producing T cells displayed cytotoxic T lymphocyte characteristics. Furthermore, individuals with psoriasis had significantly higher frequencies of circulating GAS-reactive T cells, enriched for markers of activation, cytolytic potential, and tissue association. In addition to responding to GAS, subsets of expanded GAS-reactive T cell clones/lines were found to be autoreactive, which included the recognition of the self-lipid antigen lysophosphatidylcholine. CD8+ T cell clones/lines produced cytolytic mediators and lysed infected CD1a-expressing cells. Furthermore, we established cutaneous models of GAS infection in a humanized CD1a transgenic mouse model and identified enhanced and prolonged local and systemic inflammation, with resolution through a psoriasis-like phenotype. Together, these findings link GAS infection to the CD1a pathway and show that GAS infection promotes the proliferation and activation of CD1a-autoreactive T cells, with relevance to post-streptococcal disease, including the pathogenesis and treatment of psoriasis.
Conflict of interest statement
G.O., C.S.H., and Y.-L.C. hold patent filings related to CD1a. G.O., J.N., and Y.-L.C. have relevant research collaborations with Janssen. G.O. has received relevant research support from UCB, Janssen administered through the University of Oxford. All other authors declare that they have no competing interests.
Figures






References
-
- Griffiths CEM, Armstrong AW, Gudjonsson JE, Barker J. Psoriasis. Lancet. 2021;397:1301–1315. - PubMed
-
- Whyte HJ, Baughman RD. Acute Guttate Psoriasis and Streptococcal Infection. Arch Dermatol. 1964;89:350–356. - PubMed
-
- Gudjonsson JE, Thorarinsson AM, Sigurgeirsson B, Kristinsson KG, Valdimarsson H. Streptococcal throat infections and exacerbation of chronic plaque psoriasis: a prospective study. Br J Dermatol. 2003;149:530–534. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Research Materials