Chronic heat stress induces renal fibrosis and mitochondrial dysfunction in laying hens
- PMID: 37268977
- PMCID: PMC10239180
- DOI: 10.1186/s40104-023-00878-5
Chronic heat stress induces renal fibrosis and mitochondrial dysfunction in laying hens
Abstract
Background: Heat stress in laying hens negatively affects egg production and shell quality by disrupting the homeostasis of plasma calcium and phosphorus levels. Although the kidney plays an important role in calcium and phosphorus homeostasis, evidence regarding the effect of heat stress on renal injury in laying hens is yet to be elucidated. Therefore, the aim of this study was to evaluate the effects of chronic heat stress on renal damage in hens during laying periods.
Methods: A total of 16 white-leghorn laying hens (32 weeks old) were randomly assigned to two groups (n = 8). One group was exposed to chronic heat stress (33 °C for 4 weeks), whereas the other group was maintained at 24 °C.
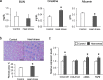
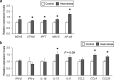
Results: Chronic heat exposure significantly increased plasma creatinine and decreased plasma albumin levels (P < 0.05). Heat exposure also increased renal fibrosis and the transcription levels of fibrosis-related genes (COLA1A1, αSMA, and TGF-β) in the kidney. These results suggest that renal failure and fibrosis were induced by chronic heat exposure in laying hens. In addition, chronic heat exposure decreased ATP levels and mitochondrial DNA copy number (mtDNA-CN) in renal tissue, suggesting that renal mitochondrial dysfunction occurs under conditions of heat stress. Damaged mitochondria leak mtDNAs into the cytosol and mtDNA leakage may activate the cyclic GMP-AMP synthase (cGAS) stimulator of interferon genes (STING) signaling pathway. Our results showed that chronic heat exposure activated the cGAS-STING pathway as indicated by increased expression of MDA5, STING, IRF7, MAVS, and NF-κB levels. Furthermore, the expression of pro-inflammatory cytokines (IL-12) and chemokines (CCL4 and CCL20) was upregulated in heat-stressed hens.
Conclusions: These results suggest that chronic heat exposure induces renal fibrosis and mitochondrial damage in laying hens. Mitochondrial damage by heat stress may activate the mtDNA-cGAS-STING signaling and cause subsequent inflammation, which contributes to the progression of renal fibrosis and dysfunction.
Keywords: Heat stress; Laying hens; Mitochondrial DNA; Renal fibrosis; cGAS-STING.
© 2023. The Author(s).
Conflict of interest statement
The authors declare that they have no competing interests.
Figures
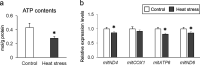
References
-
- Al-Saffar AA, Rose SP. Ambient temperature and the egg laying characteristics of laying fowl. Worlds Poult Sci J. 2002;58:317–331. doi: 10.1079/WPS20020025. - DOI
-
- Allahverdi A, Feizi A, Takhtfooladi HA, Nikpiran H. Effects of heat stress on acid-base imbalance, plasma calcium concentration, egg production and egg quality in commercial layers. Glob Vet. 2013;10:203–207.
-
- Scott TA, Balnave D. Comparison between concentrated complete diets and self-selection for feeding sexually-maturing pullets at hot and cold temperatures. Br Poult Sci. 1988;29:613–626. doi: 10.1080/00071668808417088. - DOI
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

