Application of Nano-Delivery Systems in Lymph Nodes for Tumor Immunotherapy
- PMID: 37269391
- PMCID: PMC10239433
- DOI: 10.1007/s40820-023-01125-2
Application of Nano-Delivery Systems in Lymph Nodes for Tumor Immunotherapy
Abstract
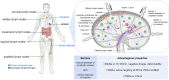
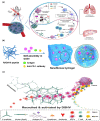
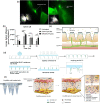
Immunotherapy has become a promising research "hotspot" in cancer treatment. "Soldier" immune cells are not uniform throughout the body; they accumulate mostly in the immune organs such as the spleen and lymph nodes (LNs), etc. The unique structure of LNs provides the microenvironment suitable for the survival, activation, and proliferation of multiple types of immune cells. LNs play an important role in both the initiation of adaptive immunity and the generation of durable anti-tumor responses. Antigens taken up by antigen-presenting cells in peripheral tissues need to migrate with lymphatic fluid to LNs to activate the lymphocytes therein. Meanwhile, the accumulation and retaining of many immune functional compounds in LNs enhance their efficacy significantly. Therefore, LNs have become a key target for tumor immunotherapy. Unfortunately, the nonspecific distribution of the immune drugs in vivo greatly limits the activation and proliferation of immune cells, which leads to unsatisfactory anti-tumor effects. The efficient nano-delivery system to LNs is an effective strategy to maximize the efficacy of immune drugs. Nano-delivery systems have shown beneficial in improving biodistribution and enhancing accumulation in lymphoid tissues, exhibiting powerful and promising prospects for achieving effective delivery to LNs. Herein, the physiological structure and the delivery barriers of LNs were summarized and the factors affecting LNs accumulation were discussed thoroughly. Moreover, developments in nano-delivery systems were reviewed and the transformation prospects of LNs targeting nanocarriers were summarized and discussed.
Keywords: Cancer therapy; Immunotherapy; Lymph nodes; Nano-delivery systems.
© 2023. The Author(s).
Conflict of interest statement
The authors declare no interest conflict. They have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Figures








Similar articles
-
Lymph node-targeting nanovaccines for cancer immunotherapy.J Control Release. 2022 Nov;351:102-122. doi: 10.1016/j.jconrel.2022.09.015. Epub 2022 Sep 20. J Control Release. 2022. PMID: 36115556 Review.
-
Recent Advances of Emerging Spleen-Targeting Nanovaccines for Immunotherapy.Adv Healthc Mater. 2023 Sep;12(23):e2300351. doi: 10.1002/adhm.202300351. Epub 2023 Jun 15. Adv Healthc Mater. 2023. PMID: 37289567 Review.
-
Cascade Cytosol Delivery of Dual-Sensitive Micelle-Tailored Vaccine for Enhancing Cancer Immunotherapy.ACS Appl Mater Interfaces. 2018 Nov 7;10(44):37797-37811. doi: 10.1021/acsami.8b09946. Epub 2018 Oct 25. ACS Appl Mater Interfaces. 2018. PMID: 30360105
-
In Situ Tumor Vaccine for Lymph Nodes Delivery and Cancer Therapy Based on Small Size Nanoadjuvant.Small. 2023 Aug;19(33):e2301041. doi: 10.1002/smll.202301041. Epub 2023 Apr 20. Small. 2023. PMID: 37078903
-
Chemical Strategies to Enhance the Therapeutic Efficacy of Toll-like Receptor Agonist Based Cancer Immunotherapy.Acc Chem Res. 2020 Oct 20;53(10):2081-2093. doi: 10.1021/acs.accounts.0c00337. Epub 2020 Sep 23. Acc Chem Res. 2020. PMID: 32966047
Cited by
-
Engineered upconversion nanoparticles for breast cancer theranostics.Theranostics. 2025 Jul 25;15(16):8259-8319. doi: 10.7150/thno.116153. eCollection 2025. Theranostics. 2025. PMID: 40860139 Free PMC article. Review.
-
A Stable Open-Shell Conjugated Diradical Polymer with Ultra-High Photothermal Conversion Efficiency for NIR-II Photo-Immunotherapy of Metastatic Tumor.Nanomicro Lett. 2023 Nov 20;16(1):21. doi: 10.1007/s40820-023-01219-x. Nanomicro Lett. 2023. PMID: 37982963 Free PMC article.
-
Nanomedicine-induced programmed cell death enhances tumor immunotherapy.J Adv Res. 2024 Aug;62:199-217. doi: 10.1016/j.jare.2023.09.018. Epub 2023 Sep 22. J Adv Res. 2024. PMID: 37743016 Free PMC article. Review.
-
Fabrication and functional validation of a hybrid biomimetic nanovaccine (HBNV) against Kit K641E -mutant melanoma.Bioact Mater. 2024 Dec 27;46:347-364. doi: 10.1016/j.bioactmat.2024.12.023. eCollection 2025 Apr. Bioact Mater. 2024. PMID: 39834347 Free PMC article.
-
Antigen/adjuvant-free liposome induces adjuvant effects for enhancing cancer immunotherapy.Exploration (Beijing). 2024 Jul 17;5(2):20230115. doi: 10.1002/EXP.20230115. eCollection 2025 Apr. Exploration (Beijing). 2024. PMID: 40395757 Free PMC article.
References
-
- Xie X, Song T, Feng Y, Zhang H, Yang G, et al. Nanotechnology-based multifunctional vaccines for cancer immunotherapy. Chem. Eng. J. 2022;437:135505. doi: 10.1016/j.cej.2022.135505. - DOI
Publication types
LinkOut - more resources
Full Text Sources
