Arginine vasopressin potentiates inspiratory bursting in hypoglossal motoneurons of neonatal mice
- PMID: 37269889
- PMCID: PMC10443434
- DOI: 10.1016/j.resp.2023.104087
Arginine vasopressin potentiates inspiratory bursting in hypoglossal motoneurons of neonatal mice
Abstract
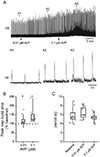
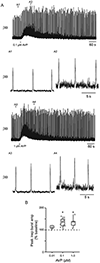
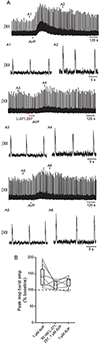
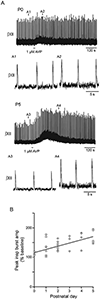
Vasopressin (AVP) acts as a neurotransmitter and its activity can potentiate respiratory activity. Hypoglossal (XII) motoneurons that innervate the tongue express V1a vasopressin receptors, which are excitatory. Therefore, we hypothesized that V1a receptor activation at XII motoneurons would potentiate inspiratory bursting. We developed this study to determine whether AVP can potentiate inspiratory bursting in rhythmic medullary slice preparations in neonatal (postnatal, P0-5) mice. Bath or local application of AVP potentiated inspiratory bursting compared to baseline XII inspiratory burst amplitude. Antagonizing V1a receptors revealed significant attenuation of the AVP-mediated potentiation of inspiratory bursting, while antagonism of oxytocin receptors (at which AVP has similar binding affinity) revealed a trend to attenuate AVP-mediated potentiation of inspiratory bursting. Finally, we discovered that the AVP-mediated potentiation of inspiratory bursting increases significantly with postnatal maturation from P0-5. Overall, these data support that AVP potentiates inspiratory bursting directly at XII motoneurons.
Keywords: Anti-diuretic hormone; Breathing; Hypoglossal; Inspiratory pattern; Motor neurons; Neuromodulation; Oxytocin; Respiration; V1a.
Copyright © 2023 Elsevier B.V. All rights reserved.
Conflict of interest statement
Declaration of Competing Interest The authors declare no competing interests.
Figures


Similar articles
-
A muscarinic, GIRK channel-mediated inhibition of inspiratory-related XII nerve motor output emerges in early postnatal development in mice.J Appl Physiol (1985). 2023 Nov 1;135(5):1041-1052. doi: 10.1152/japplphysiol.00042.2023. Epub 2023 Sep 28. J Appl Physiol (1985). 2023. PMID: 37767557 Free PMC article.
-
The vasopressin-induced excitation of hypoglossal and facial motoneurons in young rats is mediated by V1a but not V1b receptors, and is independent of intracellular calcium signalling.Eur J Neurosci. 2006 Sep;24(6):1565-74. doi: 10.1111/j.1460-9568.2006.05038.x. Eur J Neurosci. 2006. PMID: 17004920
-
Modulation of hypoglossal motoneuron excitability by NK1 receptor activation in neonatal mice in vitro.J Physiol. 2001 Jul 15;534(Pt. 2):447-64. doi: 10.1111/j.1469-7793.2001.00447.x. J Physiol. 2001. PMID: 11454963 Free PMC article.
-
Synaptic control of motoneuron excitability in rodents: from months to milliseconds.Clin Exp Pharmacol Physiol. 2000 Jan-Feb;27(1-2):120-5. doi: 10.1046/j.1440-1681.2000.03202.x. Clin Exp Pharmacol Physiol. 2000. PMID: 10696540 Review.
-
Retracted: Control of hypoglossal pre-inspiratory discharge.Exp Physiol. 2020 Aug;105(8):1232-1255. doi: 10.1113/EP087329. Epub 2020 Jul 16. Exp Physiol. 2020. Retraction in: Exp Physiol. 2020 Nov;105(11):1972. doi: 10.1113/EP089093. PMID: 32539192 Retracted. Review.
References
-
- Adachi T, Huxtable AG, Fang X, and Funk GD. Substance P modulation of hypoglossal motoneuron excitability during development: changing balance between conductances. J Neurophysiol 104: 854–872, 2010. - PubMed
-
- Bankir L, Bichet DG, and Morgenthaler NG. Vasopressin: physiology, assessment and osmosensation. J Intern Med 282: 284–297, 2017. - PubMed
-
- Barberis C, Mouillac B, and Durroux T. Structural bases of vasopressin/oxytocin receptor function. J Endocrinol 156: 223–229, 1998. - PubMed
-
- Bayliss DA, Viana F, and Berger AJ. Mechanisms underlying excitatory effects of thyrotropin-releasing hormone on rat hypoglossal motoneurons in vitro. J Neurophysiol 68: 1733–1745, 1992. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

