Concentration of Phosphatidylserine Influence Rates of Insulin Aggregation and Toxicity of Amyloid Aggregates In Vitro
- PMID: 37279439
- PMCID: PMC10401552
- DOI: 10.1021/acschemneuro.3c00277
Concentration of Phosphatidylserine Influence Rates of Insulin Aggregation and Toxicity of Amyloid Aggregates In Vitro
Abstract
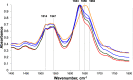
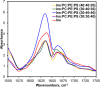
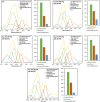
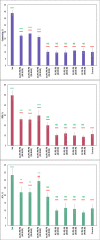
Phosphatidylserine (PS) is a negatively charged lipid that plays a critically important role in cell apoptosis. Under physiological conditions, PS is localized on the cytosolic side of plasma membranes via ATP-dependent flippase-mediated transport. A decrease in the ATP levels in the cell, which is taken place upon pathological processes, results in the increase in PS concentration on the exterior part of the cell membranes. PS on the outer membrane surfaces attracts and activates phagocytes, which trigger cell apoptosis. This programed irreversible cell death is observed upon the progressive neurodegeneration, a hallmark of numerous amyloid associated pathologies, such as diabetes type 2 and Alzheimer's disease. In this study, we investigate the extent to which the rates of protein aggregation, which occurs upon amyloid pathologies, can be altered by the concentration of PS in large unilamellar vesicles (LUVs). We found that with an increase in the concentration of PS from 20 to 40% relative to the concentration of phosphatidylcholine and phosphatidylethanolamine, the rate of insulin aggregation, protein linked to diabetes type 2, and injection amyloidosis drastically increased. Furthermore, the concentration of PS in LUVs determined the secondary structure of protein aggregates formed in their presence. We also found that these structurally different aggregates exerted distinctly different cell toxicities. These findings suggest that a substantial decrease in cell viability, which is likely to take place upon aging, results in the increase in the concentration of PS in the outer plasma membranes, where it triggers the irreversible self-assembly of amyloidogenic proteins, which, in turn, causes the progressive neurodegeneration.
Keywords: AFM-IR; fibrils; insulin; oligomers; phosphatidylserine.
Conflict of interest statement
The authors declare no competing financial interest.
Figures


Similar articles
-
Elucidating the Role of Lipids in the Aggregation of Amyloidogenic Proteins.Acc Chem Res. 2023 Nov 7;56(21):2898-2906. doi: 10.1021/acs.accounts.3c00386. Epub 2023 Oct 12. Acc Chem Res. 2023. PMID: 37824095 Free PMC article.
-
The degree of unsaturation of fatty acids in phosphatidylserine alters the rate of insulin aggregation and the structure and toxicity of amyloid aggregates.FEBS Lett. 2022 Jun;596(11):1424-1433. doi: 10.1002/1873-3468.14369. Epub 2022 May 13. FEBS Lett. 2022. PMID: 35510803 Free PMC article.
-
Lipids uniquely alter secondary structure and toxicity of lysozyme aggregates.FASEB J. 2022 Oct;36(10):e22543. doi: 10.1096/fj.202200841R. FASEB J. 2022. PMID: 36094052 Free PMC article.
-
Heterogeneous amyloid-formed ion channels as a common cytotoxic mechanism: implications for therapeutic strategies against amyloidosis.Cell Biochem Biophys. 2002;36(2-3):191-207. doi: 10.1385/CBB:36:2-3:191. Cell Biochem Biophys. 2002. PMID: 12139405 Review.
-
How our bodies fight amyloidosis: effects of physiological factors on pathogenic aggregation of amyloidogenic proteins.Arch Biochem Biophys. 2015 Feb 15;568:46-55. doi: 10.1016/j.abb.2015.01.007. Epub 2015 Jan 20. Arch Biochem Biophys. 2015. PMID: 25615529 Review.
Cited by
-
Elucidating the Role of Lipids in the Aggregation of Amyloidogenic Proteins.Acc Chem Res. 2023 Nov 7;56(21):2898-2906. doi: 10.1021/acs.accounts.3c00386. Epub 2023 Oct 12. Acc Chem Res. 2023. PMID: 37824095 Free PMC article.
-
Tubulin-binding region alters tau-lipid interactions and changes toxicity of tau fibrils formed in the presence of phosphatidylserine lipids.Protein Sci. 2024 Jul;33(7):e5078. doi: 10.1002/pro.5078. Protein Sci. 2024. PMID: 38895991 Free PMC article.
-
Insulin resistance as the molecular link between diabetes and Alzheimer's disease.World J Diabetes. 2024 Jul 15;15(7):1430-1447. doi: 10.4239/wjd.v15.i7.1430. World J Diabetes. 2024. PMID: 39099819 Free PMC article. Review.
-
Elucidation of cytotoxicity of α-Synuclein fibrils on immune cells.Biochim Biophys Acta Proteins Proteom. 2025 Feb 1;1873(2):141061. doi: 10.1016/j.bbapap.2024.141061. Epub 2024 Dec 16. Biochim Biophys Acta Proteins Proteom. 2025. PMID: 39694308 Free PMC article.
-
Under Heparin-Free Conditions Unsaturated Phospholipids Inhibit the Aggregation of 1N4R and 2N4R Tau.J Phys Chem Lett. 2024 Aug 22;15(33):8577-8583. doi: 10.1021/acs.jpclett.4c01718. Epub 2024 Aug 14. J Phys Chem Lett. 2024. PMID: 39140785 Free PMC article.
References
-
- Chen S. W.; Drakulic S.; Deas E.; Ouberai M.; Aprile F. A.; Arranz R.; Ness S.; Roodveldt C.; Guilliams T.; De-Genst E. J.; Klenerman D.; Wood N. W.; Knowles T. P.; Alfonso C.; Rivas G.; Abramov A. Y.; Valpuesta J. M.; Dobson C. M.; Cremades N. Structural characterization of toxic oligomers that are kinetically trapped during alpha-synuclein fibril formation. Proc. Natl. Acad. Sci. U.S.A. 2015, 112, E1994–E2003. 10.1073/pnas.1421204112. - DOI - PMC - PubMed
-
- Sandberg A.; Luheshi L. M.; Sollvander S.; Pereira de Barros T.; Macao B.; Knowles T. P.; Biverstal H.; Lendel C.; Ekholm-Petterson F.; Dubnovitsky A.; Lannfelt L.; Dobson C. M.; Hard T. Stabilization of neurotoxic Alzheimer amyloid-beta oligomers by protein engineering. Proc. Natl. Acad. Sci. U.S.A. 2010, 107, 15595–15600. 10.1073/pnas.1001740107. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

