Possible involvement of Interleukin-17A in the deterioration of prepulse inhibition on acoustic startle response in mice
- PMID: 37280178
- PMCID: PMC10496063
- DOI: 10.1002/npr2.12351
Possible involvement of Interleukin-17A in the deterioration of prepulse inhibition on acoustic startle response in mice
Abstract
Aim: Proinflammatory cytokines such as interleukin-6 (IL-6) and IL-17A have been implicated in the pathophysiology of schizophrenia which often shows sensorimotor gating abnormalities. This study aimed to examine whether a proinflammatory cytokine, IL-17A, induces impairment in sensorimotor gating in mice. We also examined whether IL-17A administration affects GSK3α/β protein level or phosphorylation in the striatum.
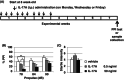
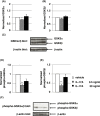
Methods: Recombinant mouse IL-17A (low-dose: 0.5 ng/mL and high-dose: 50 ng/mL with 10 μL/g mouse body weight, respectively) or vehicle was intraperitoneally administered into C57BL/6 male mice 10 times in 3 weeks (sub-chronic administration). Prepulse inhibition test using acoustic startle stimulus was conducted 4 weeks after the final IL-17A administration. We evaluated the effect of IL-17A administration on protein level or phosphorylation of GSK3α/β in the striatum by using Western blot analysis.
Results: Administration of IL-17A induced significant PPI deterioration. Low-dose of IL-17A administration significantly decreased both GSK3α (Ser21) and GSK3β (Ser9) phosphorylation in mouse striatum. There was no significant alteration of GSK3α/β protein levels except for GSK3α in low-dose IL-17A administration group.
Conclusion: We demonstrated for the first time that sub-chronic IL-17A administration induced PPI disruption and that IL-17A administration resulted in decreased phosphorylation of GSKα/β at the striatum. These results suggest that IL-17A could be a target molecule in the prevention and treatment of sensorimotor gating abnormalities observed in schizophrenia.
Keywords: GSK3; IL-17A; mouse; prepulse inhibition; sensorimotor gating; striatum.
© 2023 The Authors. Neuropsychopharmacology Reports published by John Wiley & Sons Australia, Ltd on behalf of The Japanese Society of Neuropsychopharmacology.
Conflict of interest statement
The authors declare that they have no conflicts of interest.
Figures
References
-
- Potvin S, Stip E, Sepehry AA, Gendron A, Bah R, Kouassi E. Inflammatory cytokine alterations in schizophrenia: a systematic quantitative review. Biol Psychiatry. 2008;63(8):801–8. - PubMed
-
- Sasayama D, Wakabayashi C, Hori H, Teraishi T, Hattori K, Ota M, et al. Association of plasma IL‐6 and soluble IL‐6 receptor levels with the Asp358Ala polymorphism of the IL‐6 receptor gene in schizophrenic patients. J Psychiatr Res. 2011;45(11):1439–44. - PubMed
-
- Sasayama D, Hattori K, Wakabayashi C, Teraishi T, Hori H, Ota M, et al. Increased cerebrospinal fluid interleukin‐6 levels in patients with schizophrenia and those with major depressive disorder. J Psychiatr Res. 2013;47(3):401–6. - PubMed
-
- Roytblat L, Rachinsky M, Fisher A, Greemberg L, Shapira Y, Douvdevani A, et al. Raised interleukin‐6 levels in obese patients. Obes Res. 2000;8(9):673–5. - PubMed
-
- Cano P, Cardinali DP, Rios‐Lugo MJ, Fernandez‐Mateos MP, Reyes Toso CF, Esquifino AI. Effect of a high‐fat diet on 24‐hour pattern of circulating adipocytokines in rats. Obesity (Silver Spring). 2009;17(10):1866–71. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

