Physiological activation of human and mouse bitter taste receptors by bile acids
- PMID: 37286811
- PMCID: PMC10247784
- DOI: 10.1038/s42003-023-04971-3
Physiological activation of human and mouse bitter taste receptors by bile acids
Abstract
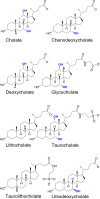
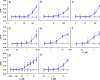
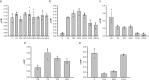
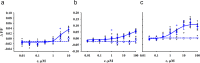
Beside the oral cavity, bitter taste receptors are expressed in several non-gustatory tissues. Whether extra-oral bitter taste receptors function as sensors for endogenous agonists is unknown. To address this question, we devised functional experiments combined with molecular modeling approaches to investigate human and mouse receptors using a variety of bile acids as candidate agonists. We show that five human and six mouse receptors are responsive to an array of bile acids. Moreover, their activation threshold concentrations match published data of bile acid concentrations in human body fluids, suggesting a putative physiological activation of non-gustatory bitter receptors. We conclude that these receptors could serve as sensors for endogenous bile acid levels. These results also indicate that bitter receptor evolution may not be driven solely by foodstuff or xenobiotic stimuli, but also depend on endogenous ligands. The determined bitter receptor activation profiles of bile acids now enable detailed physiological model studies.
© 2023. The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures


Similar articles
-
Membrane-bound chemoreception of bitter bile acids and peptides is mediated by the same subset of bitter taste receptors.Cell Mol Life Sci. 2024 May 15;81(1):217. doi: 10.1007/s00018-024-05202-6. Cell Mol Life Sci. 2024. PMID: 38748186 Free PMC article.
-
Reengineering the ligand sensitivity of the broadly tuned human bitter taste receptor TAS2R14.Biochim Biophys Acta Gen Subj. 2018 Oct;1862(10):2162-2173. doi: 10.1016/j.bbagen.2018.07.009. Epub 2018 Jul 19. Biochim Biophys Acta Gen Subj. 2018. PMID: 30009876
-
Comprehensive Analysis of Mouse Bitter Taste Receptors Reveals Different Molecular Receptive Ranges for Orthologous Receptors in Mice and Humans.J Biol Chem. 2016 Jul 15;291(29):15358-77. doi: 10.1074/jbc.M116.718544. Epub 2016 May 20. J Biol Chem. 2016. PMID: 27226572 Free PMC article.
-
International Union of Basic and Clinical Pharmacology. CXVII: Taste 2 receptors-Structures, functions, activators, and blockers.Pharmacol Rev. 2025 Jan;77(1):100001. doi: 10.1124/pharmrev.123.001140. Epub 2024 Nov 22. Pharmacol Rev. 2025. PMID: 39952694 Review.
-
The bitterness of genitourinary infections: Properties, ligands of genitourinary bitter taste receptors and mechanisms linking taste sensing to inflammatory processes in the genitourinary tract.Eur J Obstet Gynecol Reprod Biol. 2020 Apr;247:101-110. doi: 10.1016/j.ejogrb.2020.02.015. Epub 2020 Feb 14. Eur J Obstet Gynecol Reprod Biol. 2020. PMID: 32088528 Review.
Cited by
-
Bitter taste receptors as sensors of gut luminal contents.Nat Rev Gastroenterol Hepatol. 2025 Jan;22(1):39-53. doi: 10.1038/s41575-024-01005-z. Epub 2024 Oct 28. Nat Rev Gastroenterol Hepatol. 2025. PMID: 39468215 Review.
-
BitterMasS: Predicting Bitterness from Mass Spectra.J Agric Food Chem. 2024 May 8;72(18):10537-10547. doi: 10.1021/acs.jafc.3c09767. Epub 2024 Apr 30. J Agric Food Chem. 2024. PMID: 38685906 Free PMC article.
-
The Remarkable Diversity of Vertebrate Bitter Taste Receptors: Recent Advances in Genomic and Functional Studies.Int J Mol Sci. 2024 Nov 25;25(23):12654. doi: 10.3390/ijms252312654. Int J Mol Sci. 2024. PMID: 39684366 Free PMC article. Review.
-
Membrane-bound chemoreception of bitter bile acids and peptides is mediated by the same subset of bitter taste receptors.Cell Mol Life Sci. 2024 May 15;81(1):217. doi: 10.1007/s00018-024-05202-6. Cell Mol Life Sci. 2024. PMID: 38748186 Free PMC article.
-
Emerging paradigms for target discovery of traditional medicines: A genome-wide pan-GPCR perspective.Innovation (Camb). 2025 Jan 17;6(3):100774. doi: 10.1016/j.xinn.2024.100774. eCollection 2025 Mar 3. Innovation (Camb). 2025. PMID: 40098666 Free PMC article. Review.
References
-
- Behrens, M. & Meyerhof, W. in Chemosensory Transduction (eds Frank Zufall & S. D. Munger) 227–244 (Academic Press, 2016).
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

