A versatile laser-induced porcine model of outer retinal and choroidal degeneration for preclinical testing
- PMID: 37288665
- PMCID: PMC10393234
- DOI: 10.1172/jci.insight.157654
A versatile laser-induced porcine model of outer retinal and choroidal degeneration for preclinical testing
Abstract
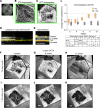
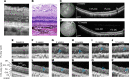
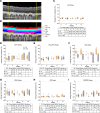
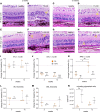
Over 30 million people worldwide suffer from untreatable vision loss and blindness associated with childhood-onset and age-related eye diseases caused by photoreceptor (PR), retinal pigment epithelium (RPE), and choriocapillaris (CC) degeneration. Recent work suggests that RPE-based cell therapy may slow down vision loss in late stages of age-related macular degeneration (AMD), a polygenic disease induced by RPE atrophy. However, accelerated development of effective cell therapies is hampered by the lack of large-animal models that allow testing safety and efficacy of clinical doses covering the human macula (20 mm2). We developed a versatile pig model to mimic different types and stages of retinal degeneration. Using an adjustable power micropulse laser, we generated varying degrees of RPE, PR, and CC damage and confirmed the damage by longitudinal analysis of clinically relevant outcomes, including analyses by adaptive optics and optical coherence tomography/angiography, along with automated image analysis. By imparting a tunable yet targeted damage to the porcine CC and visual streak - with a structure similar to the human macula - this model is optimal for testing cell and gene therapies for outer retinal diseases including AMD, retinitis pigmentosa, Stargardt, and choroideremia. The amenability of this model to clinically relevant imaging outcomes will facilitate faster translation to patients.
Keywords: Gene therapy; Ophthalmology; Retinopathy; Stem cell transplantation; Stem cells.
Conflict of interest statement
Figures




References
Publication types
MeSH terms
Supplementary concepts
LinkOut - more resources
Full Text Sources
Medical
Research Materials

