Assessing the comparability of cycle threshold values derived from five external quality assessment rounds for omicron nucleic acid testing
- PMID: 37291570
- PMCID: PMC10249569
- DOI: 10.1186/s12985-023-02032-z
Assessing the comparability of cycle threshold values derived from five external quality assessment rounds for omicron nucleic acid testing
Abstract
Background: A variety of open-system real-time reverse transcriptase polymerase chain reaction (RT-PCR) assays for several acute respiratory syndrome coronavirus 2 are currently in use. This study aimed to ensure the quality of omicron nucleic acid testing and to assess the comparability of cycle threshold (Ct) values derived from RT-PCR.
Methods: Five external quality assessment (EQA) rounds using the omicron virus-like particles were organized between February 2022 and June 2022.
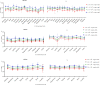
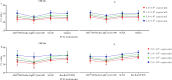
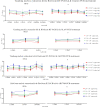
Results: A total of 1401 qualitative EQA reports have been collected. The overall positive percentage agreement was 99.72%, the negative percentage agreement was 99.75%, and the percent agreement was 99.73%. This study observed a significant variance in Ct values derived from different test systems. There was a wide heterogeneity in PCR efficiency among different RT-PCR kits and inter-laboratories.
Conclusion: There was strong concordance among laboratories performing qualitative omicron nucleic acid testing. Ct values from qualitative RT-PCR tests should not be used for clinical or epidemiological decision-making to avoid the potential for misinterpretation of the results.
Keywords: Comparability; Cycle threshold; Omicron; Qualitative test; RT-PCR; SARS-CoV-2.
© 2023. The Author(s).
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures

Similar articles
-
Performance of SARS-CoV-2 nucleic acid amplification testing in Austria as measured by external quality assessment schemes during 3 years of the COVID-19 pandemic: an observational retrospective study.Lancet Microbe. 2023 Dec;4(12):e1015-e1023. doi: 10.1016/S2666-5247(23)00286-0. Epub 2023 Nov 15. Lancet Microbe. 2023. PMID: 37979591
-
A comparative study on the efficiency of commercial reverse transcriptase-Polymerase chain reaction kits for the detection of severe acute respiratory syndrome coronavirus 2 infections.Indian J Public Health. 2022 Jul-Sep;66(3):276-281. doi: 10.4103/ijph.ijph_2042_21. Indian J Public Health. 2022. PMID: 36149104
-
Variability of cycle threshold values in an external quality assessment scheme for detection of the SARS-CoV-2 virus genome by RT-PCR.Clin Chem Lab Med. 2020 Dec 16;59(5):987-994. doi: 10.1515/cclm-2020-1602. Print 2021 Apr 27. Clin Chem Lab Med. 2020. PMID: 33554519
-
Cycle Threshold Values from Severe Acute Respiratory Syndrome Coronavirus-2 Reverse Transcription-Polymerase Chain Reaction Assays: Interpretation and Potential Use Cases.Clin Lab Med. 2022 Jun;42(2):237-248. doi: 10.1016/j.cll.2022.02.003. Epub 2022 Feb 21. Clin Lab Med. 2022. PMID: 35636824 Free PMC article. Review.
-
Laboratory Considerations for Reporting Cycle Threshold Value in COVID-19.EJIFCC. 2022 Aug 8;33(2):80-93. eCollection 2022 Aug. EJIFCC. 2022. PMID: 36313906 Free PMC article. Review.
Cited by
-
Cycle Threshold Values of SARS-CoV-2 RT-PCR during Outbreaks in Nursing Homes: A Retrospective Cohort Study.Epidemiologia (Basel). 2024 Oct 16;5(4):658-668. doi: 10.3390/epidemiologia5040046. Epidemiologia (Basel). 2024. PMID: 39449389 Free PMC article.
-
Population-Level SARS-CoV-2 RT-PCR Cycle Threshold Values and Their Relationships with COVID-19 Transmission and Outcome Metrics: A Time Series Analysis Across Pandemic Years.Viruses. 2025 Jan 14;17(1):103. doi: 10.3390/v17010103. Viruses. 2025. PMID: 39861892 Free PMC article.
-
Droplet Digital PCR or Real-Time PCR as a Method for Quantifying SARS-CoV-2 RNA in Plasma-Is There a Difference?Viruses. 2025 May 28;17(6):772. doi: 10.3390/v17060772. Viruses. 2025. PMID: 40573363 Free PMC article.
-
Comparison of two polymerase chain reaction assays, DaAn Gene (DaAn Gene 2019-ncov) and Cepheid (Cepheid Xpert Xpress SARS-CoV-2) for Delta and Omicron variants of SARS-CoV-2 detection in Yaoundé, Cameroon.J Public Health Afr. 2025 Jul 31;16(1):1282. doi: 10.4102/jphia.v16i1.1282. eCollection 2025. J Public Health Afr. 2025. PMID: 40799375 Free PMC article.
References
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

