This is a preprint.
Anopheles salivary apyrase regulates blood meal hemostasis and drives malaria parasite transmission
- PMID: 37292610
- PMCID: PMC10245845
- DOI: 10.1101/2023.05.22.541827
Anopheles salivary apyrase regulates blood meal hemostasis and drives malaria parasite transmission
Update in
-
Mosquito salivary apyrase regulates blood meal hemostasis and facilitates malaria parasite transmission.Nat Commun. 2024 Sep 18;15(1):8194. doi: 10.1038/s41467-024-52502-3. Nat Commun. 2024. PMID: 39294191 Free PMC article.
Abstract
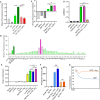
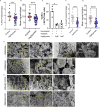
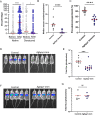
Mosquito salivary proteins play a crucial role in regulating hemostatic responses at the bite site during blood feeding. In this study, we investigate the function of Anopheles gambiae salivary apyrase (AgApyrase) in Plasmodium transmission. Our results demonstrate that salivary apyrase interacts with and activates tissue plasminogen activator, facilitating the conversion of plasminogen to plasmin, a human protein previously shown to be required for Plasmodium transmission. Microscopy imaging shows that mosquitoes ingest a substantial amount of apyrase during blood feeding which reduces coagulation in the blood meal by enhancing fibrin degradation and inhibiting platelet aggregation. Supplementation of Plasmodium infected blood with apyrase significantly enhanced Plasmodium infection in the mosquito midgut. In contrast, AgApyrase immunization inhibited Plasmodium mosquito infection and sporozoite transmission. This study highlights a pivotal role for mosquito salivary apyrase for regulation of hemostasis in the mosquito blood meal and for Plasmodium transmission to mosquitoes and to the mammal host, underscoring the potential for new strategies to prevent malaria transmission.
Conflict of interest statement
Competing interests: All data are available in the manuscript or the supplementary materials.
Figures

References
-
- WHO, World malaria report 2022. 2022.
Publication types
LinkOut - more resources
Full Text Sources
