This is a preprint.
Assortative mating and parental genetic relatedness drive the pathogenicity of variably expressive variants
- PMID: 37292616
- PMCID: PMC10246151
- DOI: 10.1101/2023.05.18.23290169
Assortative mating and parental genetic relatedness drive the pathogenicity of variably expressive variants
Update in
-
Assortative mating and parental genetic relatedness contribute to the pathogenicity of variably expressive variants.Am J Hum Genet. 2023 Dec 7;110(12):2015-2028. doi: 10.1016/j.ajhg.2023.10.015. Epub 2023 Nov 17. Am J Hum Genet. 2023. PMID: 37979581 Free PMC article.
Abstract
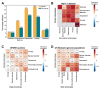
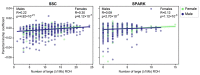
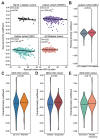
We examined more than 38,000 spouse pairs from four neurodevelopmental disease cohorts and the UK Biobank to identify phenotypic and genetic patterns in parents associated with neurodevelopmental disease risk in children. We identified correlations between six phenotypes in parents and children, including correlations of clinical diagnoses such as obsessive-compulsive disorder (R=0.31-0.49, p<0.001), and two measures of sub-clinical autism features in parents affecting several autism severity measures in children, such as bi-parental mean Social Responsiveness Scale (SRS) scores affecting proband SRS scores (regression coefficient=0.11, p=0.003). We further describe patterns of phenotypic and genetic similarity between spouses, where spouses show both within- and cross-disorder correlations for seven neurological and psychiatric phenotypes, including a within-disorder correlation for depression (R=0.25-0.72, p<0.001) and a cross-disorder correlation between schizophrenia and personality disorder (R=0.20-0.57, p<0.001). Further, these spouses with similar phenotypes were significantly correlated for rare variant burden (R=0.07-0.57, p<0.0001). We propose that assortative mating on these features may drive the increases in genetic risk over generations and the appearance of "genetic anticipation" associated with many variably expressive variants. We further identified parental relatedness as a risk factor for neurodevelopmental disorders through its inverse correlations with burden and pathogenicity of rare variants and propose that parental relatedness drives disease risk by increasing genome-wide homozygosity in children (R=0.09-0.30, p<0.001). Our results highlight the utility of assessing parent phenotypes and genotypes in predicting features in children carrying variably expressive variants and counseling families carrying these variants.
Figures


References
-
- Kvarnung M. & Nordgren A. Intellectual Disability & Rare Disorders: A Diagnostic Challenge. Adv Exp Med Biol 1031, 39–54 (2017). - PubMed
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources
