Modulation of supramolecular chirality by stepwise axial coordination in a nano-size trizinc(ii)porphyrin trimer
- PMID: 37293642
- PMCID: PMC10246700
- DOI: 10.1039/d3sc00858d
Modulation of supramolecular chirality by stepwise axial coordination in a nano-size trizinc(ii)porphyrin trimer
Abstract
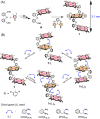
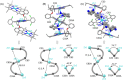
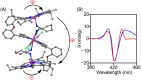
Herein, we report a chiral guest's triggered spring-like contraction and extension motions coupled with unidirectional twisting in a novel flexible and 'nano-size' achiral trizinc(ii)porphyrin trimer host upon step-wise formation of 1 : 1, 1 : 2, and 1 : 4 host-guest supramolecular complexes based on the stoichiometry of the diamine guests for the first time. During these processes, porphyrin CD responses have been induced, inverted, and amplified, and reduced, respectively, in a single molecular framework due to the change in the interporphyrin interactions and helicity. Also, the sign of the CD couplets is just the opposite between R and S substrates which suggests that the chirality is dictated solely by the stereographic projection of the chiral center. Interestingly, the long-range electronic communications between the three porphyrin rings generate trisignate CD signals that provide further information about molecular structures.
This journal is © The Royal Society of Chemistry.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures

References
-
- Taniguchi T. and Usuki T., Supramolecular Chemistry: From Molecules to Nanomaterials, John Wiley & Sons Ltd, New York, 2012
-
- Hegstrom R. A. Kondepudi D. K. Sci. Am. 1990;262:108. doi: 10.1038/scientificamerican0190-108. - DOI
-
- Bassani D. M. Lehn J.-M. Baum G. Fenske D. Angew. Chem., Int. Ed. 1997;36:1845. doi: 10.1002/anie.199718451. - DOI
LinkOut - more resources
Full Text Sources

