Uncovering diffusive states of the yeast membrane protein, Pma1, and how labeling method can change diffusive behavior
- PMID: 37294385
- PMCID: PMC10369454
- DOI: 10.1140/epje/s10189-023-00301-x
Uncovering diffusive states of the yeast membrane protein, Pma1, and how labeling method can change diffusive behavior
Abstract
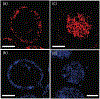
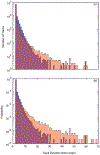
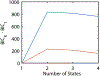
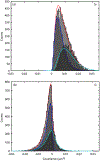
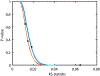
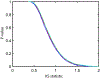
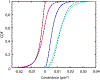
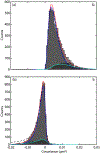
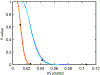
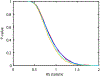
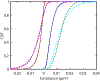
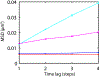
We present and analyze video-microscopy-based single-particle-tracking measurements of the budding yeast (Saccharomyces cerevisiae) membrane protein, Pma1, fluorescently labeled either by direct fusion to the switchable fluorescent protein, mEos3.2, or by a novel, light-touch, labeling scheme, in which a 5 amino acid tag is directly fused to the C-terminus of Pma1, which then binds mEos3.2. The track diffusivity distributions of these two populations of single-particle tracks differ significantly, demonstrating that labeling method can be an important determinant of diffusive behavior. We also applied perturbation expectation maximization (pEMv2) (Koo and Mochrie in Phys Rev E 94(5):052412, 2016), which sorts trajectories into the statistically optimum number of diffusive states. For both TRAP-labeled Pma1 and Pma1-mEos3.2, pEMv2 sorts the tracks into two diffusive states: an essentially immobile state and a more mobile state. However, the mobile fraction of Pma1-mEos3.2 tracks is much smaller ([Formula: see text]) than the mobile fraction of TRAP-labeled Pma1 tracks ([Formula: see text]). In addition, the diffusivity of Pma1-mEos3.2's mobile state is several times smaller than the diffusivity of TRAP-labeled Pma1's mobile state. Thus, the two different labeling methods give rise to very different overall diffusive behaviors. To critically assess pEMv2's performance, we compare the diffusivity and covariance distributions of the experimental pEMv2-sorted populations to corresponding theoretical distributions, assuming that Pma1 displacements realize a Gaussian random process. The experiment-theory comparisons for both the TRAP-labeled Pma1 and Pma1-mEos3.2 reveal good agreement, bolstering the pEMv2 approach.
© 2023. The Author(s), under exclusive licence to EDP Sciences, SIF and Springer-Verlag GmbH Germany, part of Springer Nature.
Conflict of interest statement
Figures


References
-
- Goormaghtigh E, Chadwick C, Scarborough GA, Monomers of the neurospora plasma membrane H+-ATPase catalyze efficient proton translocation. J. Biol. Chem 261, 7466–7471 (1986) - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

