Semaglutide reduces alcohol intake and relapse-like drinking in male and female rats
- PMID: 37295046
- PMCID: PMC10363436
- DOI: 10.1016/j.ebiom.2023.104642
Semaglutide reduces alcohol intake and relapse-like drinking in male and female rats
Abstract
Background: Glucagon-like peptide1 receptor (GLP-1R) agonists have been found to reduce alcohol drinking in rodents and overweight patients with alcohol use disorder (AUD). However, the probability of low semaglutide doses, an agonist with higher potency and affinity for GLP-1R, to attenuate alcohol-related responses in rodents and the underlying neuronal mechanisms is unknown.
Methods: In the intermittent access model, we examined the ability of semaglutide to decrease alcohol intake and block relapse-like drinking, as well as imaging the binding of fluorescently marked semaglutide to nucleus accumbens (NAc) in both male and female rats. The suppressive effect of semaglutide on alcohol-induced locomotor stimulation and in vivo dopamine release in NAc was tested in male mice. We evaluated effect of semaglutide on the in vivo release of dopamine metabolites (DOPAC and HVA) and gene expression of enzymes metabolising dopamine (MAOA and COMT) in male mice.
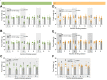
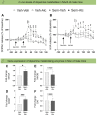
Findings: In male and female rats, acute and repeated semaglutide administration reduced alcohol intake and prevented relapse-like drinking. Moreover, fluorescently labelled semaglutide was detected in NAc of alcohol-drinking male and female rats. Further, semaglutide attenuated the ability of alcohol to cause hyperlocomotion and to elevate dopamine in NAc in male mice. As further shown in male mice, semaglutide enhanced DOPAC and HVA in NAc when alcohol was onboard and increased the gene expression of COMT and MAOA.
Interpretation: Altogether, this indicates that semaglutide reduces alcohol drinking behaviours, possibly via a reduction in alcohol-induced reward and NAc dependent mechanisms. As semaglutide also decreased body weight of alcohol-drinking rats of both sexes, upcoming clinical studies should test the plausibility that semaglutide reduces alcohol intake and body weight in overweight AUD patients.
Funding: Swedish Research Council (2019-01676), LUA/ALF (723941) from the Sahlgrenska University Hospital and the Swedish brain foundation.
Keywords: Addition; Dopamine; GLP-1; Gut-brain axis; Nucleus accumbens; Reward.
Copyright © 2023 The Author(s). Published by Elsevier B.V. All rights reserved.
Conflict of interest statement
Declaration of interests EJ has secured funding for the current project, with the support being facilitated by the University. Additionally, EJ has authored a book chapter and consequently received royalties. These financial considerations have had no bearing on the project including the design of the experiments, the analysis and interpretation of the data, and the writing of the manuscript. The remaining authors declare no conflict of interest.
Figures


References
-
- Koob G.F. Neurocircuitry of alcohol addiction: synthesis from animal models. Handb Clin Neurol. 2014;125:33–54. - PubMed
-
- Boileau I., Assaad J.M., Pihl R.O., et al. Alcohol promotes dopamine release in the human nucleus accumbens. Synapse. 2003;49(4):226–231. - PubMed
-
- Engel J.A., Fahlke C., Hulthe P., et al. Biochemical and behavioral evidence for an interaction between ethanol and calcium channel antagonists. J Neural Transm. 1988;74(3):181–193. - PubMed
-
- Imperato A., Di Chiara G. Preferential stimulation of dopamine release in the nucleus accumbens of freely moving rats by ethanol. J Pharmacol Exp Ther. 1986;239(1):219–228. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

