Assessment of Inspiratory Effort in Spontaneously Breathing COVID-19 ARDS Patients Undergoing Helmet CPAP: A Comparison between Esophageal, Transdiaphragmatic and Central Venous Pressure Swing
- PMID: 37296817
- PMCID: PMC10252717
- DOI: 10.3390/diagnostics13111965
Assessment of Inspiratory Effort in Spontaneously Breathing COVID-19 ARDS Patients Undergoing Helmet CPAP: A Comparison between Esophageal, Transdiaphragmatic and Central Venous Pressure Swing
Abstract
Introduction: The clinical features of COVID-19 are highly variable. It has been speculated that the progression across COVID-19 may be triggered by excessive inspiratory drive activation. The aim of the present study was to assess whether the tidal swing in central venous pressure (ΔCVP) is a reliable estimate of inspiratory effort.
Methods: Thirty critically ill patients with COVID-19 ARDS underwent a PEEP trial (0-5-10 cmH2O) during helmet CPAP. Esophageal (ΔPes) and transdiaphragmatic (ΔPdi) pressure swings were measured as indices of inspiratory effort. ΔCVP was assessed via a standard venous catheter. A low and a high inspiratory effort were defined as ΔPes ≤ 10 and >15 cmH2O, respectively.
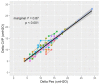
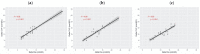
Results: During the PEEP trial, no significant changes in ΔPes (11 [6-16] vs. 11 [7-15] vs. 12 [8-16] cmH2O, p = 0.652) and in ΔCVP (12 [7-17] vs. 11.5 [7-16] vs. 11.5 [8-15] cmH2O, p = 0.918) were detected. ΔCVP was significantly associated with ΔPes (marginal R2 0.87, p < 0.001). ΔCVP recognized both low (AUC-ROC curve 0.89 [0.84-0.96]) and high inspiratory efforts (AUC-ROC curve 0.98 [0.96-1]).
Conclusions: ΔCVP is an easily available a reliable surrogate of ΔPes and can detect a low or a high inspiratory effort. This study provides a useful bedside tool to monitor the inspiratory effort of spontaneously breathing COVID-19 patients.
Keywords: ARDS; COVID-19; central venous pressure; esophageal pressure; inspiratory effort.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

References
-
- Tonelli R., Fantini R., Tabbì L., Castaniere I., Pisani L., Pellegrino M.R., Della Casa G., D’Amico R., Girardis M., Nava S., et al. Early Inspiratory Effort Assessment by Esophageal Manometry Predicts Noninvasive Ventilation Outcome in De Novo Respiratory Failure. A Pilot Study. Am. J. Respir. Crit. Care. Med. 2020;202:558–567. doi: 10.1164/rccm.201912-2512OC. - DOI - PMC - PubMed
-
- Antonelli M., Conti G., Rocco M., Bufi M., De Blasi R.A., Vivino G., Gasparetto A., Meduri G.U. A Comparison of Noninvasive Positive-Pressure Ventilation and Conventional Mechanical Ventilation in Patients with Acute Respiratory Failure. N. Engl. J. Med. 1998;339:429–435. doi: 10.1056/NEJM199808133390703. - DOI - PubMed
LinkOut - more resources
Full Text Sources

