RNA-seq analysis identifies age-dependent changes in expression of mRNAs - encoding N-glycosylation pathway enzymes in mouse gonadotropes
- PMID: 37301504
- PMCID: PMC10528389
- DOI: 10.1016/j.mce.2023.111971
RNA-seq analysis identifies age-dependent changes in expression of mRNAs - encoding N-glycosylation pathway enzymes in mouse gonadotropes
Abstract
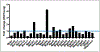
Follicle-stimulating hormone (FSH) is a glycoprotein that is assembled as a heterodimer of α/β subunits in gonadotropes. Each subunit contains two N-glycan chains. Our previous in vivo genetic studies identified that at least one N-glycan chain must be present on the FSHβ subunit for efficient FSH dimer assembly and secretion. Moreover, macroheterogeneity observed uniquely on human FSHβ results in ratiometric changes in age-specific FSH glycoforms, particularly during menopausal transition. Despite the recognition of many prominent roles of sugars on FSH including dimer assembly and secretion, serum half-life, receptor binding and signal transduction, the N-glycosylation machinery in gonadotropes has never been defined. Here, we used a mouse model in which gonadotropes are GFP-labeled in vivo and achieved rapid purification of GFP+ gonadotropes from pituitaries of female mice at reproductively young, middle, and old ages. We identified by RNA-seq analysis 52 mRNAs encoding N-glycosylation pathway enzymes expressed in 3- and 8-10-month-old mouse gonadotropes. We hierarchically mapped and localized the enzymes to distinct subcellular organelles within the N-glycosylation biosynthetic pathway. Of the 52 mRNAs, we found 27 mRNAs are differentially expressed between the 3- and 8-10-month old mice. We subsequently selected 8 mRNAs which showed varying changes in expression for confirmation of abundance in vivo via qPCR analysis, using more expanded aging time points with distinct 8-month and 14-month age groups. Real time qPCR analysis indicated dynamic changes in expression of N-glycosylation pathway enzyme-encoding mRNAs across the life span. Notably, computational analysis predicted the promoters of genes encoding these 8 mRNAs contain multiple high probability binding sites for estrogen receptor-1 and progesterone receptor. Collectively, our studies define the N-glycome and identify age-specific dynamic changes in mRNAs encoding N-glycosylation pathway enzymes in mouse gonadotropes. Our studies suggest the age-related decline in ovarian steroids may regulate expression of N-glycosylation enzymes in mouse gonadotropes and explain the age-related N-glycosylation shift previously observed on human FSHβ subunit in pituitaries of women.
Keywords: Aging; FSH Glycoforms; Gonadotrope; N-glycosylation; Ovary; Pituitary.
Copyright © 2023 Elsevier B.V. All rights reserved.
Conflict of interest statement
Declaration of competing interest The authors have nothing to disclose.
Figures

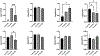

Similar articles
-
A human FSHB transgene encoding the double N-glycosylation mutant (Asn(7Δ) Asn(24Δ)) FSHβ subunit fails to rescue Fshb null mice.Mol Cell Endocrinol. 2016 May 5;426:113-24. doi: 10.1016/j.mce.2016.02.015. Epub 2016 Feb 19. Mol Cell Endocrinol. 2016. PMID: 26911932 Free PMC article.
-
Molecular characterization and expression of FSHbeta, LHbeta, and common alpha-subunit in male Atlantic halibut (Hippoglossus hippoglossus).Gen Comp Endocrinol. 2003 Apr;131(2):87-96. doi: 10.1016/s0016-6480(02)00606-8. Gen Comp Endocrinol. 2003. PMID: 12679085
-
Evaluation of in vivo bioactivities of recombinant hypo- (FSH21/18) and fully- (FSH24) glycosylated human FSH glycoforms in Fshb null mice.Mol Cell Endocrinol. 2016 Dec 5;437:224-236. doi: 10.1016/j.mce.2016.08.031. Epub 2016 Aug 22. Mol Cell Endocrinol. 2016. PMID: 27561202 Free PMC article.
-
Biased signaling by human follicle-stimulating hormone variants.Pharmacol Ther. 2025 Apr;268:108821. doi: 10.1016/j.pharmthera.2025.108821. Epub 2025 Feb 15. Pharmacol Ther. 2025. PMID: 39961417 Review.
-
Regulation of gonadotropin subunit genes in tilapia.Comp Biochem Physiol B Biochem Mol Biol. 2001 Jun;129(2-3):489-502. doi: 10.1016/s1096-4959(01)00345-1. Comp Biochem Physiol B Biochem Mol Biol. 2001. PMID: 11399484 Review.
Cited by
-
Oocyte quality is enhanced by hypoglycosylated FSH through increased cell-to-cell interaction during mouse follicle development.Development. 2023 Nov 15;150(22):dev202170. doi: 10.1242/dev.202170. Epub 2023 Nov 10. Development. 2023. PMID: 37870089 Free PMC article.
-
Glycosylation in aging and neurodegenerative diseases.Acta Biochim Biophys Sin (Shanghai). 2024 Aug 15;56(8):1208-1220. doi: 10.3724/abbs.2024136. Acta Biochim Biophys Sin (Shanghai). 2024. PMID: 39225075 Free PMC article. Review.
-
Identification of FSH-regulated and estrous stage-specific transcriptional networks in mouse ovaries.Proc Natl Acad Sci U S A. 2025 Feb 18;122(7):e2411977122. doi: 10.1073/pnas.2411977122. Epub 2025 Feb 10. Proc Natl Acad Sci U S A. 2025. PMID: 39928863 Free PMC article.
References
-
- 2019. Babraham Institute (2019). FastQC: A quality control tool for high throughput sequence data https://www.bioinformatics.babraham.ac.uk/projects/fastqc/.
-
- Akama TO, Nakagawa H, Wong NK, Sutton-Smith M, Dell A, Morris HR, Nakayama J, Nishimura S, Pai A, Moremen KW, Marth JD and Fukuda MN, 2006. Essential and mutually compensatory roles of {alpha}-mannosidase II and {alpha}-mannosidase IIx in N-glycan processing in vivo in mice, Proc Natl Acad Sci U S A. 103, 8983–8. - PMC - PubMed
-
- Anobile CJ, Talbot JA, McCann SJ, Padmanabhan V and Robertson WR, 1998. Glycoform composition of serum gonadotrophins through the normal menstrual cycle and in the post-menopausal state, Mol Hum Reprod. 4, 631–9. - PubMed
-
- Bishop LA, Nguyen TV and Schofield PR, 1995. Both of the beta-subunit carbohydrate residues of follicle-stimulating hormone determine the metabolic clearance rate and in vivo potency, Endocrinology. 136, 2635–40. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

