LncRNA PCBP1-AS1 induces osteoporosis by sponging miR-126-5p/PAK2 axis
- PMID: 37306572
- PMCID: PMC10259264
- DOI: 10.1302/2046-3758.126.BJR-2022-0324.R1
LncRNA PCBP1-AS1 induces osteoporosis by sponging miR-126-5p/PAK2 axis
Abstract
Aims: Long non-coding RNAs (lncRNAs) act as crucial regulators in osteoporosis (OP). Nonetheless, the effects and potential molecular mechanism of lncRNA PCBP1 Antisense RNA 1 (PCBP1-AS1) on OP remain largely unclear. The aim of this study was to explore the role of lncRNA PCBP1-AS1 in the pathogenesis of OP.
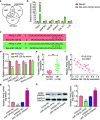
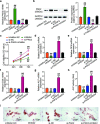
Methods: Using quantitative real-time polymerase chain reaction (qRT-PCR), osteogenesis-related genes (alkaline phosphatase (ALP), osteocalcin (OCN), osteopontin (OPN), and Runt-related transcription factor 2 (RUNX2)), PCBP1-AS1, microRNA (miR)-126-5p, group I Pak family member p21-activated kinase 2 (PAK2), and their relative expression levels were determined. Western blotting was used to examine the expression of PAK2 protein. Cell Counting Kit-8 (CCK-8) assay was used to measure cell proliferation. To examine the osteogenic differentiation, Alizarin red along with ALP staining was used. RNA immunoprecipitation assay and bioinformatics analysis, as well as a dual-luciferase reporter, were used to study the association between PCBP1-AS1, PAK2, and miR-126-5p.
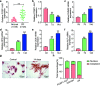
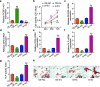
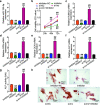
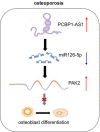
Results: The expression of PCBP1-AS1 was pre-eminent in OP tissues and decreased throughout the development of human bone marrow-derived mesenchymal stem cells (hBMSCs) into osteoblasts. PCBP1-AS1 knockdown and overexpression respectively promoted and suppressed hBMSC proliferation and osteogenic differentiation capacity. Mechanistically, PCBP1-AS1 sponged miR-126-5p and consequently targeted PAK2. Inhibiting miR-126-5p significantly counteracted the beneficial effects of PCBP1-AS1 or PAK2 knockdown on hBMSCs' ability to differentiate into osteoblasts.
Conclusion: PCBP1-AS1 is responsible for the development of OP and promotes its progression by inducing PAK2 expression via competitively binding to miR-126-5p. PCBP1-AS1 may therefore be a new therapeutic target for OP patients.
© 2023 Author(s) et al.
Conflict of interest statement
There are no competing interests, according to the author.
Figures

Similar articles
-
Super-Enhancer-Associated Long Non-Coding RNA LINC01485 Promotes Osteogenic Differentiation of Human Bone Marrow Mesenchymal Stem Cells by Regulating MiR-619-5p/RUNX2 Axis.Front Endocrinol (Lausanne). 2022 May 19;13:846154. doi: 10.3389/fendo.2022.846154. eCollection 2022. Front Endocrinol (Lausanne). 2022. PMID: 35663324 Free PMC article.
-
LncRNA LINC00205 stimulates osteoporosis and contributes to spinal fracture through the regulation of the miR-26b-5p/KMT2C axis.BMC Musculoskelet Disord. 2023 Apr 4;24(1):262. doi: 10.1186/s12891-023-06136-z. BMC Musculoskelet Disord. 2023. PMID: 37016415 Free PMC article.
-
FGD5-AS1 facilitates the osteogenic differentiation of human bone marrow-derived mesenchymal stem cells via targeting the miR-506-3p/BMP7 axis.J Orthop Surg Res. 2021 Nov 12;16(1):665. doi: 10.1186/s13018-021-02694-x. J Orthop Surg Res. 2021. PMID: 34772438 Free PMC article.
-
LINC01089 governs the miR-1287-5p/HSPA4 axis to negatively regulate osteogenic differentiation of mesenchymal stem cells.Bone Joint Res. 2024 Dec 16;13(12):779-789. doi: 10.1302/2046-3758.1312.BJR-2023-0272.R2. Bone Joint Res. 2024. PMID: 39679709 Free PMC article.
-
Silencing of OIP5-AS1 Protects Endothelial Cells From ox-LDL-Triggered Injury by Regulating KLF5 Expression via Sponging miR-135a-5p.Front Cardiovasc Med. 2021 Mar 12;8:596506. doi: 10.3389/fcvm.2021.596506. eCollection 2021. Front Cardiovasc Med. 2021. PMID: 33778018 Free PMC article.
Cited by
-
Salvianic acid A promotes osteogenic differentiation of bone marrow mesenchymal stem cells in senile osteoporosis through bromodomain-containing protein 4/Ariadne RBR E3 ubiquitin-protein ligase 1/Rubicon axis.J Tradit Complement Med. 2025 Feb 7;15(4):434-445. doi: 10.1016/j.jtcme.2025.02.003. eCollection 2025 Jul. J Tradit Complement Med. 2025. PMID: 40677544 Free PMC article.
-
The role of palmitoylation modifications in the regulation of bone cell function, bone homeostasis, and osteoporosis.Bone Joint Res. 2025 May 9;14(5):420-433. doi: 10.1302/2046-3758.145.BJR-2024-0259.R2. Bone Joint Res. 2025. PMID: 40341006 Free PMC article.
-
lncRNA Ubr5 promotes BMSCs apoptosis and inhibits their proliferation and osteogenic differentiation in weightless bone loss.Front Cell Dev Biol. 2025 Apr 2;13:1543929. doi: 10.3389/fcell.2025.1543929. eCollection 2025. Front Cell Dev Biol. 2025. PMID: 40241795 Free PMC article.
-
Knockdown of MIF-AS1 Inhibits Pain and Inflammation in Lumbar Disc Herniation by Modulating the miR-185-5p/VEGFA Axis.Global Spine J. 2025 Jun 3:21925682251336711. doi: 10.1177/21925682251336711. Online ahead of print. Global Spine J. 2025. PMID: 40460143 Free PMC article.
-
Cyclooxygenase-2 negatively regulates osteogenic differentiation in murine bone marrow mesenchymal stem cells via the FOXO3a/p27kip1 pathway.Bone Joint Res. 2025 May 8;14(5):407-419. doi: 10.1302/2046-3758.145.BJR-2024-0262.R2. Bone Joint Res. 2025. PMID: 40335058 Free PMC article.
References
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

