Reduced Cerebellar BDNF Availability Affects Postnatal Differentiation and Maturation of Granule Cells in a Mouse Model of Cholesterol Dyshomeostasis
- PMID: 37314654
- PMCID: PMC10415459
- DOI: 10.1007/s12035-023-03435-3
Reduced Cerebellar BDNF Availability Affects Postnatal Differentiation and Maturation of Granule Cells in a Mouse Model of Cholesterol Dyshomeostasis
Abstract
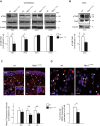
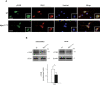
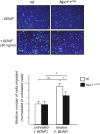
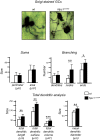
Niemann-Pick type C1 (NPC1) disease is a lysosomal lipid storage disorder due to mutations in the NPC1 gene resulting in the accumulation of cholesterol within the endosomal/lysosomal compartments. The prominent feature of the disorder is the progressive Purkinje cell degeneration leading to ataxia.In a mouse model of NPC1 disease, we have previously demonstrated that impaired Sonic hedgehog signaling causes defective proliferation of granule cells (GCs) and abnormal cerebellar morphogenesis. Studies conducted on cortical and hippocampal neurons indicate a functional interaction between Sonic hedgehog and brain-derived neurotrophic factor (BDNF) expression, leading us to hypothesize that BDNF signaling may be altered in Npc1 mutant mice, contributing to the onset of cerebellar alterations present in NPC1 disease before the appearance of signs of ataxia.We characterized the expression/localization patterns of the BDNF and its receptor, tropomyosin-related kinase B (TrkB), in the early postnatal and young adult cerebellum of the Npc1nmf164 mutant mouse strain.In Npc1nmf164 mice, our results show (i) a reduced expression of cerebellar BDNF and pTrkB in the first 2 weeks postpartum, phases in which most GCs complete the proliferative/migrative program and begin differentiation; (ii) an altered subcellular localization of the pTrkB receptor in GCs, both in vivo and in vitro; (iii) reduced chemotactic response to BDNF in GCs cultured in vitro, associated with impaired internalization of the activated TrkB receptor; (iv) an overall increase in dendritic branching in mature GCs, resulting in impaired differentiation of the cerebellar glomeruli, the major synaptic complex between GCs and mossy fibers.
Keywords: Cerebellar glomerulus; Cerebellum; Niemann-Pick type C1 disease; Npc1; TrkB receptor.
© 2023. The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures


Similar articles
-
Developmental delay in motor skill acquisition in Niemann-Pick C1 mice reveals abnormal cerebellar morphogenesis.Acta Neuropathol Commun. 2016 Sep 1;4(1):94. doi: 10.1186/s40478-016-0370-z. Acta Neuropathol Commun. 2016. PMID: 27586038 Free PMC article.
-
Shortened primary cilium length and dysregulated Sonic hedgehog signaling in Niemann-Pick C1 disease.Hum Mol Genet. 2017 Jun 15;26(12):2277-2289. doi: 10.1093/hmg/ddx118. Hum Mol Genet. 2017. PMID: 28379564
-
Endosomal dysfunction contributes to cerebellar deficits in spinocerebellar ataxia type 6.Elife. 2023 Dec 12;12:RP90510. doi: 10.7554/eLife.90510. Elife. 2023. PMID: 38084749 Free PMC article.
-
The Cerebellum in Niemann-Pick C1 Disease: Mouse Versus Man.Cerebellum. 2023 Feb;22(1):102-119. doi: 10.1007/s12311-021-01347-3. Epub 2022 Jan 18. Cerebellum. 2023. PMID: 35040097 Free PMC article. Review.
-
BDNF and Cerebellar Ataxia.Curr Drug Res Rev. 2024;16(3):300-307. doi: 10.2174/2589977515666230811093021. Curr Drug Res Rev. 2024. PMID: 37609676 Review.
Cited by
-
Cerebellum in neurodegenerative diseases: Advances, challenges, and prospects.iScience. 2024 Oct 18;27(11):111194. doi: 10.1016/j.isci.2024.111194. eCollection 2024 Nov 15. iScience. 2024. PMID: 39555407 Free PMC article. Review.
References
-
- Pentchev PG, Brady RO, Blanchette-Mackie EJ, Vanier MT, Carstea ED, Parker CC, Goldin E, Roff CF. The Niemann-Pick C lesion and its relationship to the intracellular distribution and utilization of LDL cholesterol. Biochim Biophys Acta. 1994;1225:235–243. doi: 10.1016/0925-4439(94)90001-9. - DOI - PubMed
-
- Palladino G, Loizzo S, Fortuna A, Canterini S, Palombi F, Erickson RP, Mangia F, Fiorenza MT. Visual evoked potentials of Niemann-Pick type C1 mice reveal an impairment of the visual pathway that is rescued by 2-hydroxypropyl-ß-cyclodextrin. Orphanet J Rare Dis. 2015;10:133. doi: 10.1186/s13023-015-0348-0. - DOI - PMC - PubMed
-
- Boyle BR, Melli SE, Altreche RS, Padron ZM, Fak Y, Kim S, Vasquez MD, Carone DM, Carone BR, Soto I. NPC1 deficiency impairs cerebellar postnatal development of microglia and climbing fiber refinement in a mouse model of Niemann-Pick disease type C. Development. 2020;147:dev189019. doi: 10.1242/dev.189019. - DOI - PMC - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

