Avatrombopag, a promising novel thrombopoietin receptor agonist for refractory/relapsed/intolerant non-severe aplastic anemia: a phase 2 single-arm clinical trial
- PMID: 37318085
- PMCID: PMC10274520
- DOI: 10.1080/07853890.2023.2224044
Avatrombopag, a promising novel thrombopoietin receptor agonist for refractory/relapsed/intolerant non-severe aplastic anemia: a phase 2 single-arm clinical trial
Abstract
Introduction: The therapeutic options for thrombocytopenia in non-severe aplastic anaemia (NSAA) are limited. Avatrombopag (AVA) is prescribed for thrombocytopenic diseases but not for NSAA.
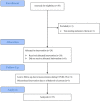
Methods: Herein, we conducted a phase 2, non-randomized, single-arm trial to explore the efficacy and safety of AVA in refractory/relapsed/intolerant NSAA. AVA dose was initiated at 20 mg/d and titrated to a maximum of 60 mg/d. The primary endpoint was the haematological response at 3 months.
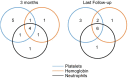
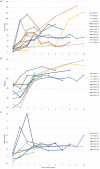
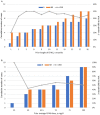
Results: Twenty-five patients were analyzed. The overall response rate (ORR) at 3 months was 56% (14/25), with 12% (3/25) achieving a complete response (CR). At a median follow-up of 7 (3-10) months, the OR and CR rates were 52% and 20%, respectively. Responders had a shorter duration of diagnosis of AVA administration than non-responders (10 (6-80) vs 37 (6-480) months, p = 0.027) and belonged to the relapsed/intolerant NSAA type (71% vs 27%, p = 0.047); 44% (8/18) patients previously treated with eltrombopag before enrollment responded at 3 months, with an average prior eltrombopag dose of median 72.5 (50-100) mg/d and an average AVA dose for a response of median 43.5 (20-60) mg/d. 3-month ORR had no significant correlation with eltrombopag exposure (p = 0.09), prior eltrombopag length (R2=0.11), or cumulative eltrombopag dose (R2=0.30). Only one patient relapsed after stopping AVA for 1 month. No serious AVA-related side effects or clone evolution were detected.
Conclusion: AVA is effective and well-tolerated in NSAA patients who are refractory, relapsed, or intolerant to CsA/tacrolimus ± eltrombopag. Earlier treatment and relapsed/intolerant AA may show a better short-term response rate. More studies are needed to define the optimal dose and the long-term efficacy (NCT04728789).
Keywords: Non-severe aplastic anemia; avatrombopag; efficacy; side effects; switch.
Conflict of interest statement
No potential conflict of interest was reported by the author(s).
Figures
Similar articles
-
Avatrombopag as alternative therapy for severe aplastic anemia patients who are intolerant or unresponsive to eltrombopag.Front Immunol. 2024 Jul 24;15:1393829. doi: 10.3389/fimmu.2024.1393829. eCollection 2024. Front Immunol. 2024. PMID: 39114665 Free PMC article.
-
Comparison of eltrombopag and avatrombopag in the treatment of refractory/relapsed aplastic anemia: a single-center retrospective study in China.Ther Adv Hematol. 2023 Sep 14;14:20406207231191310. doi: 10.1177/20406207231191310. eCollection 2023. Ther Adv Hematol. 2023. PMID: 37719987 Free PMC article.
-
Efficacy and safety of avatrombopag in combination with immunosuppressive therapy in treatment-naïve and relapsed/refractory severe aplastic anaemia: protocol for the DIAAMOND-Ava-FIRST and DIAAMOND-Ava-NEXT Bayesian Optimal Phase II trials.BMJ Open. 2024 Jan 18;14(1):e076246. doi: 10.1136/bmjopen-2023-076246. BMJ Open. 2024. PMID: 38238183 Free PMC article.
-
Eltrombopag: a review of its use in patients with severe aplastic anaemia.Drugs. 2015 Apr;75(5):525-31. doi: 10.1007/s40265-015-0363-4. Drugs. 2015. PMID: 25700916 Review.
-
Eltrombopag in Immune Thrombocytopenia, Aplastic Anemia, and Myelodysplastic Syndrome: From Megakaryopoiesis to Immunomodulation.Drugs. 2019 Aug;79(12):1305-1319. doi: 10.1007/s40265-019-01159-0. Drugs. 2019. PMID: 31292909 Review.
Cited by
-
Cyclosporine plus avatrombopag versus cyclosporine monotherapy for first-line treatment of elderly patients with transfusion-dependent non-severe aplastic anemia: a single center retrospective study.Ann Med. 2024 Dec;56(1):2424451. doi: 10.1080/07853890.2024.2424451. Epub 2024 Nov 12. Ann Med. 2024. PMID: 39529509 Free PMC article.
-
Avatrombopag for adults with early versus chronic immune thrombocytopenia.Am J Hematol. 2024 Feb;99(2):155-162. doi: 10.1002/ajh.27080. Epub 2023 Dec 8. Am J Hematol. 2024. PMID: 38063420 Free PMC article.
-
Modified Delphi panel consensus recommendations for management of severe aplastic anemia.Blood Adv. 2024 Aug 13;8(15):3946-3960. doi: 10.1182/bloodadvances.2023011642. Blood Adv. 2024. PMID: 38669341 Free PMC article.
-
Effective treatment of refractory aplastic anemia with romiplostim after failure of multiple thrombopoietin receptor agonists: a single-center retrospective study.Ann Med. 2025 Dec;57(1):2514791. doi: 10.1080/07853890.2025.2514791. Epub 2025 Jun 19. Ann Med. 2025. PMID: 40534403 Free PMC article.
-
Exploring the Potential of Pyridine Carboxylic Acid Isomers to Discover New Enzyme Inhibitors.Drug Des Devel Ther. 2025 May 20;19:4039-4091. doi: 10.2147/DDDT.S513461. eCollection 2025. Drug Des Devel Ther. 2025. PMID: 40420948 Free PMC article. Review.
References
Publication types
MeSH terms
Substances
Associated data
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
