Recovery of valuable metals from spent lithium-ion batteries using microbial agents for bioleaching: a review
- PMID: 37323903
- PMCID: PMC10264615
- DOI: 10.3389/fmicb.2023.1197081
Recovery of valuable metals from spent lithium-ion batteries using microbial agents for bioleaching: a review
Abstract
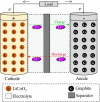
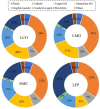
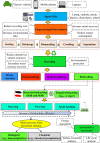
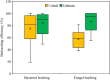
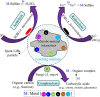
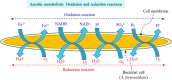
Spent lithium-ion batteries (LIBs) are increasingly generated due to their widespread use for various energy-related applications. Spent LIBs contain several valuable metals including cobalt (Co) and lithium (Li) whose supply cannot be sustained in the long-term in view of their increased demand. To avoid environmental pollution and recover valuable metals, recycling of spent LIBs is widely explored using different methods. Bioleaching (biohydrometallurgy), an environmentally benign process, is receiving increased attention in recent years since it utilizes suitable microorganisms for selective leaching of Co and Li from spent LIBs and is cost-effective. A comprehensive and critical analysis of recent studies on the performance of various microbial agents for the extraction of Co and Li from the solid matrix of spent LIBs would help for development of novel and practical strategies for effective extraction of precious metals from spent LIBs. Specifically, this review focuses on the current advancements in the application of microbial agents namely bacteria (e.g., Acidithiobacillus ferrooxidans and Acidithiobacillus thiooxidans) and fungi (e.g., Aspergillus niger) for the recovery of Co and Li from spent LIBs. Both bacterial and fungal leaching are effective for metal dissolution from spent LIBs. Among the two valuable metals, the dissolution rate of Li is higher than Co. The key metabolites which drive the bacterial leaching include sulfuric acid, while citric acid, gluconic acid and oxalic acid are the dominant metabolites in fungal leaching. The bioleaching performance depends on both biotic (microbial agents) and abiotic factors (pH, pulp density, dissolved oxygen level and temperature). The major biochemical mechanisms which contribute to metal dissolution include acidolysis, redoxolysis and complexolysis. In most cases, the shrinking core model is suitable to describe the bioleaching kinetics. Biological-based methods (e.g., bioprecipitation) can be applied for metal recovery from the bioleaching solution. There are several potential operational challenges and knowledge gaps which should be addressed in future studies to scale-up the bioleaching process. Overall, this review is of importance from the perspective of development of highly efficient and sustainable bioleaching processes for optimum resource recovery of Co and Li from spent LIBs, and conservation of natural resources to achieve circular economy.
Keywords: biohydrometallurgy; bioleaching; cathode material; circular economy; lithium and cobalt; metal recovery; spent Li-ion batteries; sustainability.
Copyright © 2023 Biswal and Balasubramanian.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures
Similar articles
-
Enhanced bioleaching of spent Li-ion batteries using A. ferrooxidans by application of external magnetic field.J Environ Manage. 2024 Sep;367:122012. doi: 10.1016/j.jenvman.2024.122012. Epub 2024 Aug 1. J Environ Manage. 2024. PMID: 39094417
-
A novel closed-loop biotechnology for recovery of cobalt from a lithium-ion battery active cathode material.Microbiology (Reading). 2024 Jul;170(7):001475. doi: 10.1099/mic.0.001475. Microbiology (Reading). 2024. PMID: 39016549 Free PMC article.
-
Improvement of Li and Mn bioleaching from spent lithium-ion batteries, using step-wise addition of biogenic sulfuric acid by Acidithiobacillus thiooxidans.Heliyon. 2024 Sep 6;10(18):e37447. doi: 10.1016/j.heliyon.2024.e37447. eCollection 2024 Sep 30. Heliyon. 2024. PMID: 39315164 Free PMC article.
-
Lithium bioleaching: An emerging approach for the recovery of Li from spent lithium ion batteries.Chemosphere. 2021 Aug;277:130196. doi: 10.1016/j.chemosphere.2021.130196. Epub 2021 Mar 6. Chemosphere. 2021. PMID: 33784558 Review.
-
A review on the recycling of spent lithium-ion batteries (LIBs) by the bioleaching approach.Chemosphere. 2021 Nov;282:130944. doi: 10.1016/j.chemosphere.2021.130944. Epub 2021 May 21. Chemosphere. 2021. PMID: 34087562 Review.
Cited by
-
Carbazole-Phosphazene Based Polymer for Efficient Extraction of Gold and Precious Elements from Electronic Waste.ACS Omega. 2024 Nov 18;9(48):47884-47892. doi: 10.1021/acsomega.4c09068. eCollection 2024 Dec 3. ACS Omega. 2024. PMID: 39651079 Free PMC article.
-
Aspects of Nickel, Cobalt and Lithium, the Three Key Elements for Li-Ion Batteries: An Overview on Resources, Demands, and Production.Materials (Basel). 2024 Sep 5;17(17):4389. doi: 10.3390/ma17174389. Materials (Basel). 2024. PMID: 39274778 Free PMC article. Review.
-
Lithium-ion battery second life: pathways, challenges and outlook.Front Chem. 2024 Apr 8;12:1358417. doi: 10.3389/fchem.2024.1358417. eCollection 2024. Front Chem. 2024. PMID: 38650673 Free PMC article. Review.
-
Recycling and Reuse of Spent LIBs: Technological Advances and Future Directions.Molecules. 2024 Jul 2;29(13):3161. doi: 10.3390/molecules29133161. Molecules. 2024. PMID: 38999113 Free PMC article. Review.
-
Bacterial acidic agents-assisted multi-elemental (Ni, Co, and Li) leaching of used lithium-ion batteries at high pulp densities.Sci Rep. 2025 May 13;15(1):16517. doi: 10.1038/s41598-025-00660-9. Sci Rep. 2025. PMID: 40360650 Free PMC article.
References
-
- Aboelazm E. A. A., Mohamed N., Ali G. A. M., Makhlouf A. S. H., Chong K. F. (2021). “Recycling of cobalt oxides electrodes from spent lithium-ion batteries by electrochemical method,” in Waste recycling technologies for nanomaterials manufacturing, eds Makhlouf A. S. H., Ali G. A. M. (Cham: Springer International Publishing; ), 91–123. 10.1007/978-3-030-68031-2_4 - DOI
-
- Ahmed A. T. A., Soni R., Ansari A. S., Lee C. Y., Kim H.-S., Im H., et al. (2022). Biowaste-derived graphitic carbon interfaced TiO2 as anode for lithium-ion battery. Surf. Interfaces 35:102404. 10.1016/j.surfin.2022.102404 - DOI
-
- Alavi N., Partovi K., Majlessi M., Rashidi M., Alimohammadi M. (2021). Bioleaching of metals from cellphones batteries by a co-fungus medium in presence of carbon materials. Bioresour. Technol. Rep. 15:100768. 10.1016/j.biteb.2021.100768 - DOI
-
- Ali H., Khan H. A., Pecht M. (2022). Preprocessing of spent lithium-ion batteries for recycling: Need, methods, and trends. Renew. Sustain. Energy Rev. 168:112809. 10.1016/j.rser.2022.112809 - DOI
-
- Alipanah M., Reed D., Thompson V., Fujita Y., Jin H. (2023). Sustainable bioleaching of lithium-ion batteries for critical materials recovery. J. Clean. Prod. 382:135274. 10.1016/j.jclepro.2022.135274 - DOI
Publication types
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

