Vertigoheel promotes rodent cognitive performance in multiple memory tests
- PMID: 37325043
- PMCID: PMC10264630
- DOI: 10.3389/fnins.2023.1183023
Vertigoheel promotes rodent cognitive performance in multiple memory tests
Abstract
Introduction: Cognitive impairment associated with old age or various brain disorders may be very disabling for affected individuals, placing their carers and public health services under considerable stress. The standard-of-care drugs produce only transient improvement of cognitive impairment in older people, so the search for novel, safe and effective therapeutics that would help to reverse or delay cognitive impairment is warranted. Repurposing pharmacological therapies with well-established safety record for additional indications is a promising recent trend in drug development. Vertigoheel (VH-04), a multicomponent drug made of Ambra grisea, Anamirta cocculus L., Conium maculatum, and Petroleum rectificatum, has been successfully used for several decades in the treatment of vertigo. Here, we investigated effects of VH-04 on cognitive performance in standard behavioral tests assessing different types of memory and explored cellular and molecular underpinnings of VH-04's biological activity.
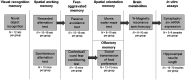
Methods: In the majority of behavioral experiments, namely in the spontaneous and rewarded alternation tests, passive avoidance test, contextual/cued fear conditioning, and social transmission of food preference, we examined the ability of single and repeated intraperitoneal administrations of VH-04 to improve cognitive parameters of mice and rats disrupted by the application of the muscarinic antagonist scopolamine. In addition, we also assessed how VH-04 affected novel object recognition and influenced performance of aged animals in Morris water maze. Furthermore, we also studied the effects of VH-04 on primary hippocampal neurons in vitro and mRNA expression of synaptophysin in the hippocampus.
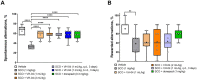
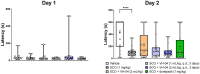
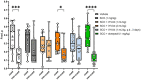
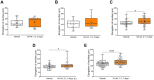
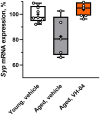
Results: Administration of VH-04 positively influenced visual recognition memory in the novel object recognition test and alleviated the impairments in spatial working memory and olfactory memory caused by the muscarinic antagonist scopolamine in the spontaneous alternation and social transmission of food preference tests. In addition, VH-04 improved retention of the spatial orientation memory of old rats in the Morris water maze. In contrast, VH-04 did not have significant effects on scopolamine-induced impairments in tests of fear-aggravated memory or rewarded alternation. Experiments in vitro showed that VH-04 stimulated neurite growth and possibly reversed the age-dependent decrease in hippocampal synaptophysin mRNA expression, which implies that VH-04 may preserve synaptic integrity in the aging brain.
Discussion: Our findings allow a cautious conclusion that in addition to its ability to alleviate manifestations of vertigo, VH-04 may be also used as a cognitive enhancer.
Keywords: Vertigoheel; forgetfulness; multicomponent drug; neurite length; olfactory memory; spatial orientation memory; spatial working memory; visual recognition memory.
Copyright © 2023 Ott, Heikkinen, Lehtimäki, Paldanius, Puoliväli, Pussinen, Andriambeloson, Huyard, Wagner, Schnack, Wahler, von Einem, von Arnim, Burmeister, Weyer and Seilheimer.
Conflict of interest statement
Charles River Discovery Services Finland Oy and Neurofit are contract research organizations. At the time of the study, KW, KO, YB, and BS were employed by Heel GmbH. TH, KL, KP, JP, and RP were employed by Charles River Discovery Services Finland Oy. EA, BH, and SW were employed by Neurofit. CS, AW, BE, and CA were employed by the Ulm University. A patent application in relation to results has been filed as International Application no. PCT/EP2012/071898 (published as WO2013/068330) resulting in national patents and patent applications EP2776131, UA112784, RU2699038, and RU2019126669.
Figures




References
-
- Akar F., Mutlu O., Celikyurt I. K., Bektas E., Tanyeri M. H., Ulak G., et al. (2014). Effects of zaprinast and rolipram on olfactory and visual memory in the social transmission of food preference and novel object recognition tests in mice. Drug Target Insights 8, 23–29. doi: 10.4137/DTI.S14813, PMID: - DOI - PMC - PubMed
-
- Buccafusco J. J. (2009). The revival of scopolamine reversal for the assessment of cognition-enhancing drugs. in Methods of behavior analysis in neuroscience. ed. Buccafusco J. J. Boca Raton, FL: CRC Press/Taylor & Francis. Available at: http://www.ncbi.nlm.nih.gov/books/NBK5233/ (Accessed September 5, 2022). - PubMed
LinkOut - more resources
Full Text Sources

