Sialidase facilitates Porphyromonas gingivalis immune evasion by reducing M1 polarization, antigen presentation, and phagocytosis of infected macrophages
- PMID: 37325520
- PMCID: PMC10266273
- DOI: 10.3389/fcimb.2023.1173899
Sialidase facilitates Porphyromonas gingivalis immune evasion by reducing M1 polarization, antigen presentation, and phagocytosis of infected macrophages
Abstract
Background: Porphyromonas gingivalis (P. gingivalis), a major pathogen of periodontitis, can evade host immune defenses. Previously, we found that P. gingivalis W83 sialidase gene mutant strain (ΔPG0352) was more easily cleared by macrophages. The aims of this study were to investigate the effects of sialidase in P. gingivalis on the polarization, antigen presentation, and phagocytosis of infected macrophages and to clarify the mechanism of P. gingivalis immune evasion.
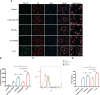
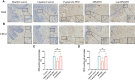
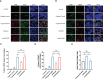
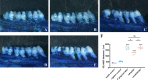
Methods: Human monocytes U937 were differentiated to macrophages and infected with P. gingivalis W83, ΔPG0352, comΔPG0352, and Escherichia coli (E. coli). The phagocytosis of macrophages was observed by transmission electron microscopy and flow cytometry. ELISA or Griess reaction were used to examine the levels of interleukin-12 (IL-12), inducible nitric oxide synthase (iNOS) and interleukin-10 (IL-10), and the expressions of CD68, CD80 and CD206 were determined by flow cytometry. The expression of major histocompatibility complex-II (MHC-II) was detected by immunofluorescence. A rat periodontitis model was established to determine the M1 and M2 polarization of macrophages.
Results: Compare with P. gingivalis W83, ΔPG0352 increased the levels of IL-12, iNOS, CD80, and MHC-II and inhibited the levels of IL-10 and CD206. Macrophages phagocytosed 75.4% of ΔPG0352 and 59.5% of P. gingivalis W83. In the rat periodontitis model, the levels of M1 and M2 macrophages in P. gingivalis W83 group were both higher than those in ΔPG0352 group, while the ratio of M1/M2 was higher in the ΔPG0352 group. Alveolar bone absorption was lower in ΔPG0352 group.
Conclusion: Sialidase facilitates P. gingivalis immune evasion by reducing M1 polarization, antigen presentation, and phagocytosis of infected macrophages.
Keywords: Porphyromonas gingivalis; macrophage; periodontitis; polarization; sialidase.
Copyright © 2023 Fan, Zheng, Chen, Lin, Wang, Shen, Pan and Li.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures


Similar articles
-
Sialidase Deficiency in Porphyromonas gingivalis Increases IL-12 Secretion in Stimulated Macrophages Through Regulation of CR3, IncRNA GAS5 and miR-21.Front Cell Infect Microbiol. 2018 Apr 5;8:100. doi: 10.3389/fcimb.2018.00100. eCollection 2018. Front Cell Infect Microbiol. 2018. PMID: 29675399 Free PMC article.
-
Differences in survival, virulence and biofilm formation between sialidase-deficient and W83 wild-type Porphyromonas gingivalis strains under stressful environmental conditions.BMC Microbiol. 2017 Aug 18;17(1):178. doi: 10.1186/s12866-017-1087-2. BMC Microbiol. 2017. PMID: 28821225 Free PMC article.
-
Unprimed, M1 and M2 Macrophages Differentially Interact with Porphyromonas gingivalis.PLoS One. 2016 Jul 6;11(7):e0158629. doi: 10.1371/journal.pone.0158629. eCollection 2016. PLoS One. 2016. PMID: 27383471 Free PMC article.
-
Macrophages: A communication network linking Porphyromonas gingivalis infection and associated systemic diseases.Front Immunol. 2022 Jul 27;13:952040. doi: 10.3389/fimmu.2022.952040. eCollection 2022. Front Immunol. 2022. PMID: 35967399 Free PMC article. Review.
-
Macrophages immunomodulation induced by Porphyromonas gingivalis and oral antimicrobial peptides.Odontology. 2023 Oct;111(4):778-792. doi: 10.1007/s10266-023-00798-w. Epub 2023 Mar 10. Odontology. 2023. PMID: 36897441 Free PMC article. Review.
Cited by
-
The emerging role of Toxoplasma gondii in periodontal diseases and underlying mechanisms.Front Immunol. 2024 Oct 3;15:1464108. doi: 10.3389/fimmu.2024.1464108. eCollection 2024. Front Immunol. 2024. PMID: 39430742 Free PMC article. Review.
-
SARS-CoV-2, periodontal pathogens, and host factors: The trinity of oral post-acute sequelae of COVID-19.Rev Med Virol. 2024 May;34(3):e2543. doi: 10.1002/rmv.2543. Rev Med Virol. 2024. PMID: 38782605 Free PMC article. Review.
-
Streptococcus suis: A Review of Its Effects on Immune Organs.Microorganisms. 2025 Jul 9;13(7):1613. doi: 10.3390/microorganisms13071613. Microorganisms. 2025. PMID: 40732121 Free PMC article. Review.
-
Macrophages in graft-versus-host disease (GVHD): dual roles as therapeutic tools and targets.Clin Exp Med. 2025 Mar 6;25(1):73. doi: 10.1007/s10238-025-01588-0. Clin Exp Med. 2025. PMID: 40048037 Free PMC article. Review.
-
Oral Pathogens' Substantial Burden on Cancer, Cardiovascular Diseases, Alzheimer's, Diabetes, and Other Systemic Diseases: A Public Health Crisis-A Comprehensive Review.Pathogens. 2024 Dec 9;13(12):1084. doi: 10.3390/pathogens13121084. Pathogens. 2024. PMID: 39770344 Free PMC article. Review.
References
-
- Castro S. A., Collighan R., Lambert P. A., Dias I. H., Chauhan P., Bland C. E., et al. . (2017). Porphyromonas gingivalis gingipains cause defective macrophage migration towards apoptotic cells and inhibit phagocytosis of primary apoptotic neutrophils. Cell Death Dis. 8, e2644. doi: 10.1038/cddis.2016.481 - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials

