The Activity of Members of the UDP-Glucuronosyltransferase Subfamilies UGT1A and UGT2B is Impaired in Patients with Liver Cirrhosis
- PMID: 37328712
- PMCID: PMC10386950
- DOI: 10.1007/s40262-023-01261-3
The Activity of Members of the UDP-Glucuronosyltransferase Subfamilies UGT1A and UGT2B is Impaired in Patients with Liver Cirrhosis
Abstract
Background and objective: The impact of liver cirrhosis on the activity of UDP-glucuronosyltransferases (UGTs) is currently not well characterized. We investigated the glucuronidation capacity and glucuronide accumulation in patients with liver cirrhosis.
Methods: We administered the Basel phenotyping cocktail (caffeine, efavirenz, flurbiprofen, omeprazole, metoprolol, midazolam) to patients with liver cirrhosis (n = 16 Child A, n = 15 Child B, n = 5 Child C) and n = 12 control subjects and obtained pharmacokinetic profiles of substrates and primary metabolites and their glucuronides.
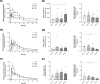
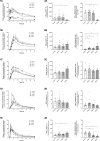
Results: Caffeine and its metabolite paraxanthine were only slightly glucuronidated. The metabolic ratio (AUCglucuronide/AUCparent, MR) was not affected for caffeine but decreased by 60% for paraxanthine glucuronide formation in Child C patients. Efavirenz was not glucuronidated whereas 8-hydroxyefavirenz was efficiently glucuronidated. The MR of 8-hydroxyefavirenz-glucuronide formation increased three-fold in Child C patients and was negatively correlated with the glomerular filtration rate. Flurbiprofen and omeprazole were not glucuronidated. 4-Hydroxyflurbiprofen and 5-hydroxyomeprazole were both glucuronidated but the corresponding MRs for glucuronide formation were not affected by liver cirrhosis. Metoprolol, but not α-hydroxymetoprolol, was glucuronidated, and the MR for metoprolol-glucuronide formation dropped by 60% in Child C patients. Both midazolam and its metabolite 1'-hydroxymidazolam underwent glucuronidation, and the corresponding MRs for glucuronide formation dropped by approximately 80% in Child C patients. No relevant glucuronide accumulation occurred in patients with liver cirrhosis.
Conclusions: Detailed analysis revealed that liver cirrhosis may affect the activity of UGTs of the UGT1A and UGT2B subfamilies according to liver function. Clinically significant glucuronide accumulation did not occur in the population investigated.
Clinical trial registration: NCT03337945.
© 2023. The Author(s).
Conflict of interest statement
Urs Duthaler, Fabio Bachmann, Agustos C. Ozbey, Kenichi Umehara, Neil Parrott, Stephen Fowler, and Stephan Krähenbühl have no conflicts of interest that are directly relevant to the content of this article.
Figures

Similar articles
-
Liver Cirrhosis Affects the Pharmacokinetics of the Six Substrates of the Basel Phenotyping Cocktail Differently.Clin Pharmacokinet. 2022 Jul;61(7):1039-1055. doi: 10.1007/s40262-022-01119-0. Epub 2022 May 16. Clin Pharmacokinet. 2022. PMID: 35570253 Free PMC article.
-
Physiologically Based Pharmacokinetic Modeling to Predict the Impact of Liver Cirrhosis on Glucuronidation via UGT1A4 and UGT2B7/2B4-A Case Study with Midazolam.Drug Metab Dispos. 2024 Jun 17;52(7):614-625. doi: 10.1124/dmd.123.001635. Drug Metab Dispos. 2024. PMID: 38653501
-
Metabolism of 1'- and 4-hydroxymidazolam by glucuronide conjugation is largely mediated by UDP-glucuronosyltransferases 1A4, 2B4, and 2B7.Drug Metab Dispos. 2010 Nov;38(11):2007-13. doi: 10.1124/dmd.110.035295. Epub 2010 Aug 16. Drug Metab Dispos. 2010. PMID: 20713656
-
Identification of uridine diphosphate glucuronosyltransferases involved in the metabolism and clearance of mycophenolic acid.Ther Drug Monit. 2000 Feb;22(1):10-3. doi: 10.1097/00007691-200002000-00002. Ther Drug Monit. 2000. PMID: 10688250 Review.
-
Species differences in drug glucuronidation: Humanized UDP-glucuronosyltransferase 1 mice and their application for predicting drug glucuronidation and drug-induced toxicity in humans.Drug Metab Pharmacokinet. 2018 Feb;33(1):9-16. doi: 10.1016/j.dmpk.2017.10.002. Epub 2017 Oct 7. Drug Metab Pharmacokinet. 2018. PMID: 29079228 Free PMC article. Review.
Cited by
-
Clinical Exploration and Physiologically Based Modelling of the Impact of Hepatic Impairment on Entrectinib Pharmacokinetics.Clin Pharmacokinet. 2025 Mar;64(3):437-451. doi: 10.1007/s40262-024-01468-y. Epub 2025 Feb 11. Clin Pharmacokinet. 2025. PMID: 39934586 Clinical Trial.
-
A Phenotyping Tool for Seven Cytochrome P450 Enzymes and Two Transporters: Application to Examine the Effects of Clopidogrel and Gemfibrozil.Clin Pharmacol Ther. 2025 Jun;117(6):1732-1742. doi: 10.1002/cpt.3610. Epub 2025 Feb 21. Clin Pharmacol Ther. 2025. PMID: 39982209 Free PMC article. Clinical Trial.
-
Simultaneously Predicting the Pharmacokinetics of CES1-Metabolized Drugs and Their Metabolites Using Physiologically Based Pharmacokinetic Model in Cirrhosis Subjects.Pharmaceutics. 2024 Feb 5;16(2):234. doi: 10.3390/pharmaceutics16020234. Pharmaceutics. 2024. PMID: 38399287 Free PMC article.
-
Pharmacometabolomics Detects Various Unreported Metoprolol Metabolites in Urine of (Potential) Living Kidney Donors and Kidney Transplant Recipients.Clin Pharmacokinet. 2025 May;64(5):779-789. doi: 10.1007/s40262-025-01502-7. Epub 2025 Apr 26. Clin Pharmacokinet. 2025. PMID: 40285825 Free PMC article.
-
Effect of Hepatic Impairment or Renal Impairment on the Pharmacokinetics of Aficamten.Clin Pharmacokinet. 2025 Mar;64(3):397-406. doi: 10.1007/s40262-025-01481-9. Epub 2025 Feb 5. Clin Pharmacokinet. 2025. PMID: 39907965 Free PMC article. Clinical Trial.
References
Publication types
MeSH terms
Substances
Associated data
LinkOut - more resources
Full Text Sources
Medical

