Bronchial epithelial cells release inflammatory markers linked to airway inflammation and remodeling in response to TLR5 ligand flagellin
- PMID: 37332524
- PMCID: PMC10276272
- DOI: 10.1016/j.waojou.2023.100786
Bronchial epithelial cells release inflammatory markers linked to airway inflammation and remodeling in response to TLR5 ligand flagellin
Abstract
Background/aims: Flagellin, which is abundant in gram-negative bacteria, including Pseudomonas, is reported to influence on inflammatory responses in various lung diseases. However, its effect on airway epithelial cells in contribution to asthma pathogenesis is not elucidated yet. We aimed to investigate the effect of TLR5 ligand flagellin on the transcriptomic profile of primary human epithelial cells and to determine the markers of airway inflammation.
Methods: Normal human bronchial epithelial (NHBE) cells were grown and differentiated in air-liquid interface (ALI) culture for 14-16 days. The cells were treated with flagellin in vitro at 10 and 100 ng/ml for 3 and 24 h. The conditioned media and cells were harvested to validate inflammatory markers involved in airway inflammation using ELISA, Western blot, and quantitative PCR methods. RNA-sequencing was performed to investigate the transcriptional response to flagellin in ALI-NHBE cells.
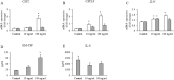
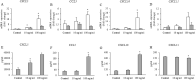
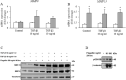
Results: Altered transcriptional responses to flagellin in differentiated bronchial epithelial cells were determined, including genes encoding chemokines, matrix metalloproteinases, and antimicrobial biomolecules. Pathway analysis of the transcriptionally responsive genes revealed enrichment of signaling pathways. Flagellin induced the mRNA expressions of proinflammatory cytokines and chemokines, and secretion of GM-CSF, CXCL5, CCL5 and CXCL10. Flagellin enhanced the protein expression of MMP-13 in TGF-β1 and TGF-β2 pretreated cell lysates and Wnt/β-catenin signaling.
Conclusions: These findings suggest that flagellin could be a potent inducer of inflammatory markers that may contribute to airway inflammation and remodeling.
Keywords: Airway; Epithelial cell; Flagellin; Inflammation; TLR5.
© 2023 The Author(s).
Figures
Similar articles
-
Pseudomonas aeruginosa stimulates tyrosine phosphorylation of and TLR5 association with the MUC1 cytoplasmic tail through EGFR activation.Inflamm Res. 2016 Mar;65(3):225-33. doi: 10.1007/s00011-015-0908-8. Epub 2015 Dec 8. Inflamm Res. 2016. PMID: 26645913 Free PMC article.
-
mTOR inhibition impacts the flagellin-augmented inflammatory and antimicrobial response of human airway epithelial cells to Pseudomonas aeruginosa.PLoS One. 2025 May 8;20(5):e0321462. doi: 10.1371/journal.pone.0321462. eCollection 2025. PLoS One. 2025. PMID: 40338861 Free PMC article.
-
Toll-like receptor 5-mediated corneal epithelial inflammatory responses to Pseudomonas aeruginosa flagellin.Invest Ophthalmol Vis Sci. 2003 Oct;44(10):4247-54. doi: 10.1167/iovs.03-0219. Invest Ophthalmol Vis Sci. 2003. PMID: 14507868
-
Plasticity of airway epithelial cell transcriptome in response to flagellin.PLoS One. 2015 Feb 10;10(2):e0115486. doi: 10.1371/journal.pone.0115486. eCollection 2015. PLoS One. 2015. PMID: 25668187 Free PMC article.
-
Human airway epithelial cells sense Pseudomonas aeruginosa infection via recognition of flagellin by Toll-like receptor 5.Infect Immun. 2005 Nov;73(11):7151-60. doi: 10.1128/IAI.73.11.7151-7160.2005. Infect Immun. 2005. PMID: 16239509 Free PMC article.
Cited by
-
Bitter taste receptor T2R14-Gαi coupling mediates innate immune responses to microbial quorum sensing molecules in cystic fibrosis.iScience. 2024 Oct 30;27(12):111286. doi: 10.1016/j.isci.2024.111286. eCollection 2024 Dec 20. iScience. 2024. PMID: 39628561 Free PMC article.
-
A cross talk between microbial metabolites and host immunity: Its relevance for allergic diseases.Clin Transl Allergy. 2024 Feb;14(2):e12339. doi: 10.1002/clt2.12339. Clin Transl Allergy. 2024. PMID: 38342758 Free PMC article. Review.
-
Toll-like receptor-mediated immune imbalance in asthma: controversies, breakthroughs, and future directions.Front Immunol. 2025 Jul 2;16:1605185. doi: 10.3389/fimmu.2025.1605185. eCollection 2025. Front Immunol. 2025. PMID: 40672946 Free PMC article. Review.
-
Epithelial Barrier Theory: The Role of Exposome, Microbiome, and Barrier Function in Allergic Diseases.Allergy Asthma Immunol Res. 2023 Nov;15(6):705-724. doi: 10.4168/aair.2023.15.6.705. Allergy Asthma Immunol Res. 2023. PMID: 37957791 Free PMC article. Review.
References
LinkOut - more resources
Full Text Sources

