Peripheral Arterial Thrombosis following Russell's Viper Bites
- PMID: 37333023
- PMCID: PMC10276757
- DOI: 10.1055/s-0043-1769625
Peripheral Arterial Thrombosis following Russell's Viper Bites
Abstract
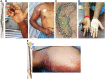
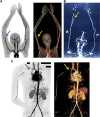
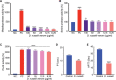
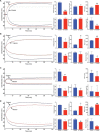
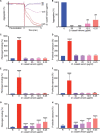
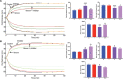
Envenomings by Russell's viper ( Daboia russelii ), a species of high medical importance in India and other Asian countries, commonly result in hemorrhage, coagulopathies, necrosis, and acute kidney injury. Although bleeding complications are frequently reported following viper envenomings, thrombotic events occur rarely (reported only in coronary and carotid arteries) with serious consequences. For the first time, we report three serious cases of peripheral arterial thrombosis following Russell's viper bites and their diagnostic, clinical management, and mechanistic insights. These patients developed occlusive thrombi in their peripheral arteries and symptoms despite antivenom treatment. In addition to clinical features, computed tomography angiography was used to diagnose arterial thrombosis and ascertain its precise locations. They were treated using thrombectomy or amputation in one case that presented with gangrenous digits. Mechanistic insights into the pathology through investigations revealed the procoagulant actions of Russell's viper venom in standard clotting tests as well as in rotational thromboelastometry analysis. Notably, Russell's viper venom inhibited agonist-induced platelet activation. The procoagulant effects of Russell's viper venom were inhibited by a matrix metalloprotease inhibitor, marimastat, although a phospholipase A 2 inhibitor (varespladib) did not show any inhibitory effects. Russell's viper venom induced pulmonary thrombosis when injected intravenously in mice and thrombi in the microvasculature and affected skeletal muscle when administered locally. These data emphasize the significance of peripheral arterial thrombosis in snakebite victims and provide awareness, mechanisms, and robust strategies for clinicians to tackle this issue in patients.
Keywords: Russell's viper; peripheral arteries; snakebite envenomation; thrombosis; venom.
The Author(s). This is an open access article published by Thieme under the terms of the Creative Commons Attribution License, permitting unrestricted use, distribution, and reproduction so long as the original work is properly cited. ( https://creativecommons.org/licenses/by/4.0/ ).
Conflict of interest statement
Conflict of Interest None declared.
Figures

References
-
- Simpson I D, Norris R L. Snakes of medical importance in India: is the concept of the “Big 4” still relevant and useful? Wilderness Environ Med. 2007;18(01):2–9. - PubMed
-
- Menon J C, Joseph J K, Whitaker R E. Venomous snake bite in India - Why do 50,000 Indians die every year? J Assoc Physicians India. 2017;65(08):78–81. - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources

