COVID-19 vaccine coverage, safety, and perceptions among patients with diabetes mellitus in China: a cross-sectional study
- PMID: 37334292
- PMCID: PMC10270113
- DOI: 10.3389/fendo.2023.1172089
COVID-19 vaccine coverage, safety, and perceptions among patients with diabetes mellitus in China: a cross-sectional study
Abstract
Aims: Diabetes mellitus (DM), one of the most common chronic diseases in China, is a risk factor for SARS-COV-2 infection and poor prognosis of COVID-19. The COVID-19 vaccine is one of the key measures to control the pandemic. However, the actual coverage of COVID-19 vaccination and associated factors remain unclear among DM patients in China. We conducted this study to investigate the COVID-19 vaccine coverage, safety, and perceptions among patients with DM in China.
Methods: A cross-sectional study of a sample of 2200 DM patients from 180 tertiary hospitals in China was performed using a questionnaire developed through the Wen Juan Xing survey platform to collect information regarding their coverage, safety, and perceptions of COVID-19 vaccination. A multinomial logistic regression analysis model was performed to determine any independent relationships with COVID-19 vaccination behavior among DM patients.
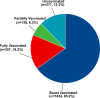
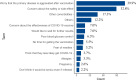
Results: In total, 1929 (87.7%) DM patients have received at least one dose COVID-19 vaccine, and 271 (12.3%) DM patients were unvaccinated. In addition, 65.2% (n = 1434) were booster vaccinated against COVID-19, while 16.2% (n = 357) were only fully vaccinated and 6.3% (n = 138) were only partially vaccinated. The prevalence of adverse effects after the first dose of vaccine, the second dose of vaccine, and the third dose of vaccine were 6.0%, 6.0%, and 4.3% respectively. Multinomial logistic regression analysis showed that DM patients complicated with immune and inflammatory diseases (partially vaccinated: OR = 0.12; fully vaccinated: OR = 0.11; booster vaccinated: OR = 0.28), diabetic nephropathy (partially vaccinated: OR = 0.23; fully vaccinated: OR = 0.50; booster vaccinated: OR = 0.30), and perceptions on the safety of COVID-19 vaccine (partially vaccinated: OR = 0.44; fully vaccinated: OR = 0.48; booster vaccinated: OR = 0.45) were all associated with the three of vaccination status.
Conclusion: This study showed that higher proportion of COVID-19 vaccine coverage among patients with DM in China. The concern about the safety of the COVID-19 vaccine affected the vaccine behavior in patients with DM. The COVID-19 vaccine was relatively safe for DM patients due to all side effects were self-limiting.
Keywords: COVID-19; China; diabetes mellitus; vaccine; vaccine uptake.
Copyright © 2023 Li, Ping, Li, Wang, Xiao, Jiang, Xue, Quan, Yao, Zheng, Chen, Li, Yu, Xu, Feng, Wang, Li and Xiao.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures

Similar articles
-
The effect of framing and communicating COVID-19 vaccine side-effect risks on vaccine intentions for adults in the UK and the USA: A structured summary of a study protocol for a randomized controlled trial.Trials. 2021 Sep 6;22(1):592. doi: 10.1186/s13063-021-05484-2. Trials. 2021. PMID: 34488843 Free PMC article.
-
Determinants of COVID-19 Vaccine Acceptance and Hesitancy: A Health Care Student-Based Online Survey in Northwest China.Front Public Health. 2022 Jan 6;9:777565. doi: 10.3389/fpubh.2021.777565. eCollection 2021. Front Public Health. 2022. PMID: 35071162 Free PMC article.
-
Safety of third SARS-CoV-2 vaccine (booster dose) during pregnancy.Am J Obstet Gynecol MFM. 2022 Jul;4(4):100637. doi: 10.1016/j.ajogmf.2022.100637. Epub 2022 Apr 7. Am J Obstet Gynecol MFM. 2022. PMID: 35398583 Free PMC article.
-
Interplay of diabetes mellitus and rheumatic diseases amidst the COVID-19 pandemic: influence on the risk of infection, outcomes, and immune responses.Clin Rheumatol. 2022 Dec;41(12):3897-3913. doi: 10.1007/s10067-022-06365-y. Epub 2022 Sep 9. Clin Rheumatol. 2022. PMID: 36076125 Free PMC article. Review.
-
SARS-CoV-2 vaccination in children and adolescents with and without type 1 diabetes mellitus.Endocrine. 2023 Nov;82(2):226-236. doi: 10.1007/s12020-023-03471-y. Epub 2023 Aug 16. Endocrine. 2023. PMID: 37587391 Free PMC article. Review.
Cited by
-
Longitudinal Evaluation of Metabolic Benefits of Inactivated COVID-19 Vaccination in Diabetic Patients in Tianjin, China.Med Sci Monit. 2025 Apr 27;31:e947450. doi: 10.12659/MSM.947450. Med Sci Monit. 2025. PMID: 40287793 Free PMC article.
-
How Does the Psychological Impact of COVID-19 Affect the Management Strategies of Individuals with Type 1 and Type 2 Diabetes? A Mixed-Method Study.Healthcare (Basel). 2024 Aug 27;12(17):1710. doi: 10.3390/healthcare12171710. Healthcare (Basel). 2024. PMID: 39273735 Free PMC article.
-
The Influence of SARS-CoV-2 Vaccination on the Mortality and Outcomes of Patients with Both Myocardial Infarction and COVID-19.Vaccines (Basel). 2024 Aug 29;12(9):983. doi: 10.3390/vaccines12090983. Vaccines (Basel). 2024. PMID: 39340015 Free PMC article.
-
Parental Factors Associated with COVID-19 Vaccine Uptake for Children over 5 Years of Age in Texas.Vaccines (Basel). 2024 May 11;12(5):526. doi: 10.3390/vaccines12050526. Vaccines (Basel). 2024. PMID: 38793777 Free PMC article.
References
-
- Lopez Bernal J, Andrews N, Gower C, Robertson C, Stowe J, Tessier E, et al. . Effectiveness of the pfizer-BioNTech and Oxford-AstraZeneca vaccines on covid-19 related symptoms, hospital admissions, and mortality in older adults in England: test negative case-control study. BMJ (Clinical Res ed) (2021) 373:n1088. doi: 10.1136/bmj.n1088 - DOI - PMC - PubMed
-
- Fiolet T, Kherabi Y, MacDonald CJ, Ghosn J, Peiffer-Smadja N. Comparing COVID-19 vaccines for their characteristics, efficacy and effectiveness against SARS-CoV-2 and variants of concern: a narrative review. Clin Microbiol infect (2022) 28(2):202–21. doi: 10.1016/j.cmi.2021.10.005 - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

